Axial partners with great founders and inventors. We invest in early-stage life sciences companies such as Appia Bio, Seranova Bio, Delix Therapeutics, Simcha Therapeutics, among others often when they are no more than an idea. We are fanatical about helping the rare inventor who is compelled to build their own enduring business. If you or someone you know has a great idea or company in life sciences, Axial would be excited to get to know you and possibly invest in your vision and company . We are excited to be in business with you - email us at info@axialvc.com
SQZ Biotech was founded in 2013 based on research from the Jensen and Langer Labs at MIT to deliver macromolecules into cells by “squeezing them.” Based on this invention, the company built a cell engineering platform (CellSqueeze) company to manufacture cell therapies. SQZ built a unique manufacturing business model with both external and internal partnerships to set the company up to build their own pipeline of medicines.
The first slide of their latest corporate presentation shows SQZ’s leadership team. The key part is that QZ is a founder-driven company.
The next slide explains the company's technology. The premise is that squeezing cells through a microfluidic channel opens up pores in the cell membrane for macromolecules like nucleic acids and proteins to enter. With this tool, the idea is that more complex cargos can be delivered to a cell without needing to use a virus.
SQZ then gets into their vision. Right now they are focused on cell therapy manufacturing. They want to get to same-day treatment in 5 years. Right now the standard turnaround time for most autologous cell therapies is 7-14 days.
SQZ is less focused on being a tools company and building out their own pipeline - with Roche highlighting their external pipeline and SQZ also building out internal products as well.
The next slide goes into milestones over the next year and a half. Focused on developing the pipeline and getting to a point-of-care device prototype.
The presentation goes into a section focused on their antigen presenting cell (APC) program with Roche for cancer vaccines.
The clinical premise of SQZ’s program is that higher levels of CD8 tumor infiltrating lymphocytes (TIL) are correlated with responses to anti-cancer drugs.
SQZ engineers APCs to activate CD8+ T-cells.
Initial preclinical data shows a ~1000x fold increase in T-cell expansion with SQZ’s APCs.
Moreover these T-cells show more specific killing toward HPV antigens.
This leads into the value proposition. Intracellular delivery of antigens into APCs might led to higher T-cell potenecy.
SQZ gives an overview on the phase 1 trial design for their lead external program for HPV.
Phase 1 data shows safety so far.
The next section touches upon using APCs for oncology.
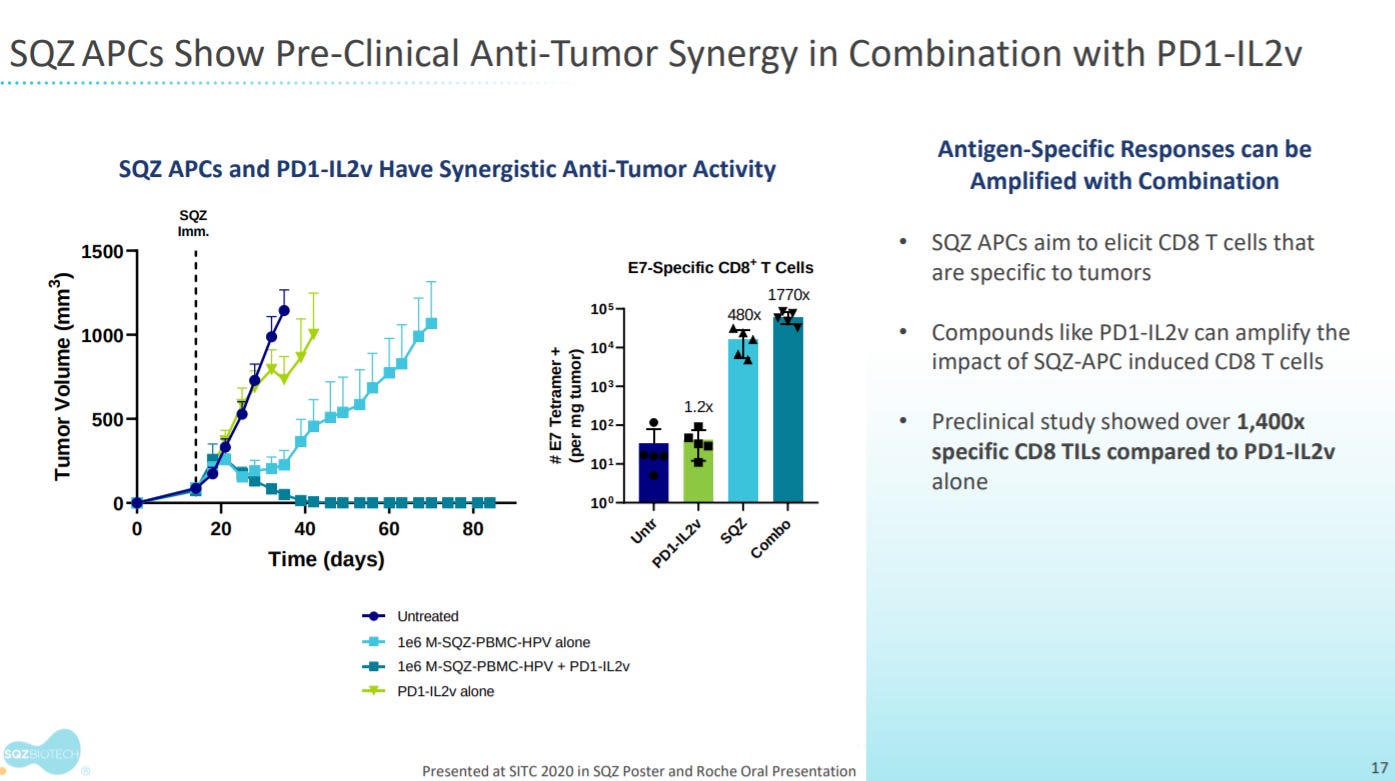
Preclinical data shows SQZ APCs show synergy with PD1-IL2v that activates IL-2R in PD-1+ T-cells.
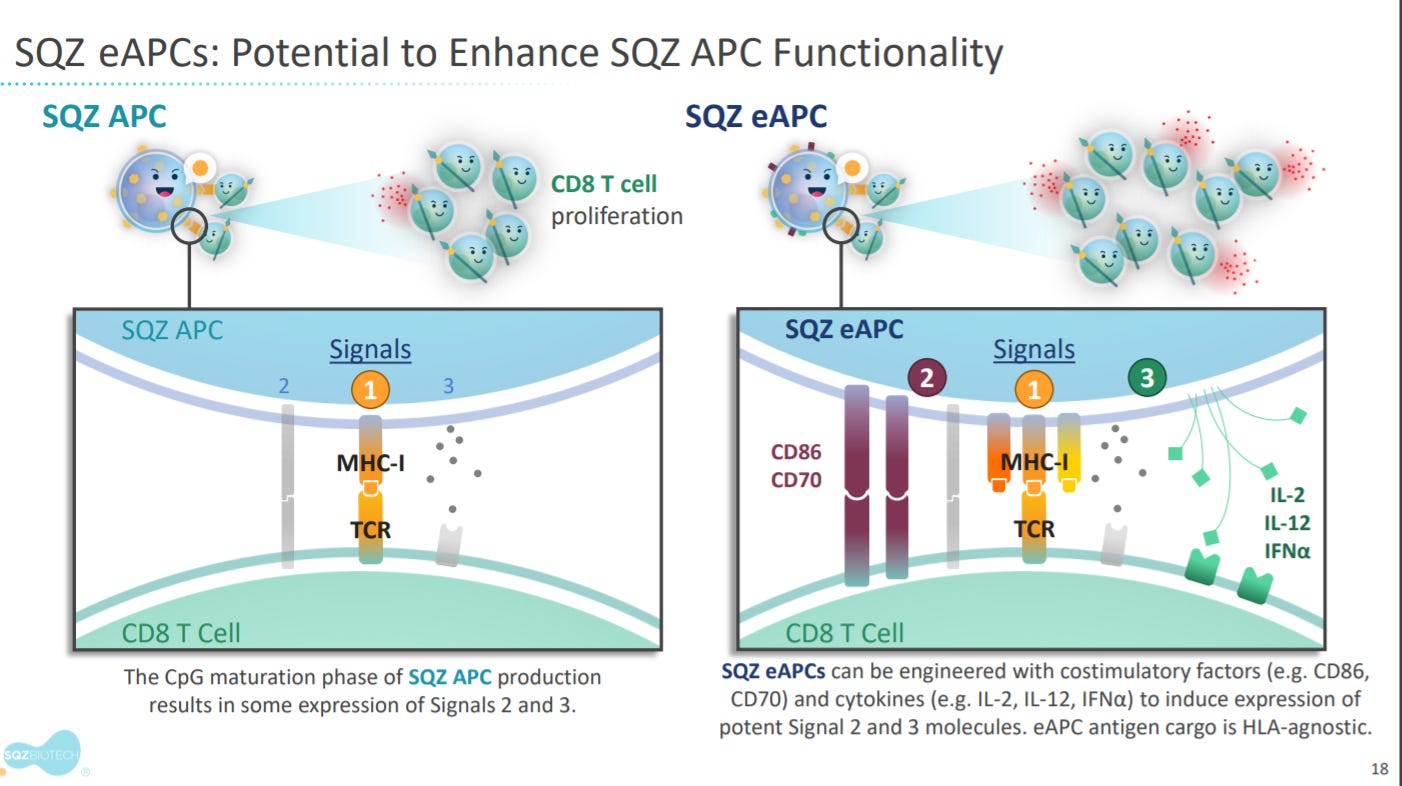
To potentiate this anti-tumor response, SQZ has also added costimulatory domains to design enhanced APCs (eAPC).
With the value proposition of overcoming HLA restrictions.
Which could expand the indications addressed by SQZ’s eAPCs.
Using KRAS mutants as an antigen did a pretty great job at eliciting CD8+ T-cells.
The next slide gives an overview on SQZ’s partnership with Roche. The deal was expanded in 2018 and given Roche’s track record, they might just end up acquiring their partner.
Manufacturing for SQZ is focused on one week turnaround times and want to get to same-day dosing.
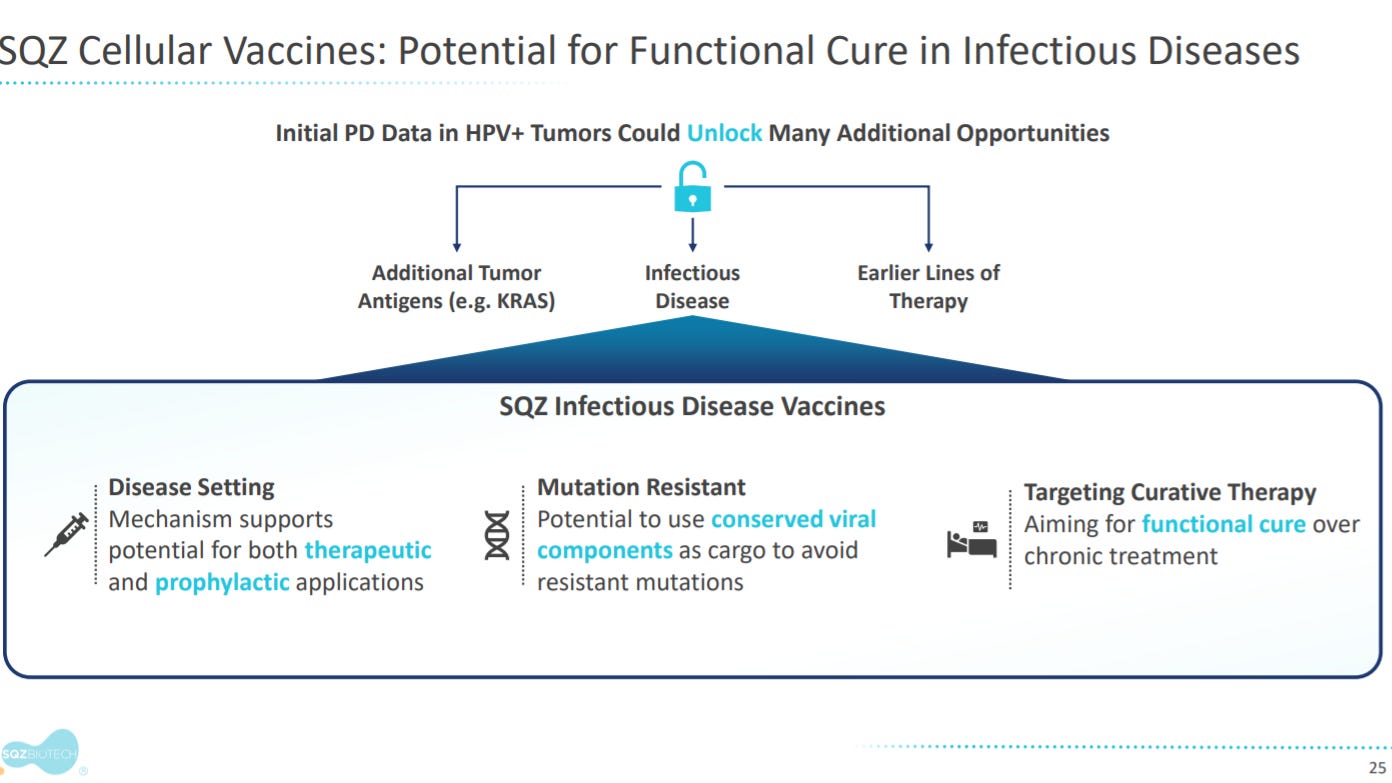
The next section gives an overview on APCs applications in infectious disease.
With early validation of APCs in HPV+ cancers, SQZ wants to enter the infectious disease vaccine field.
Their lead program here is in HBV.
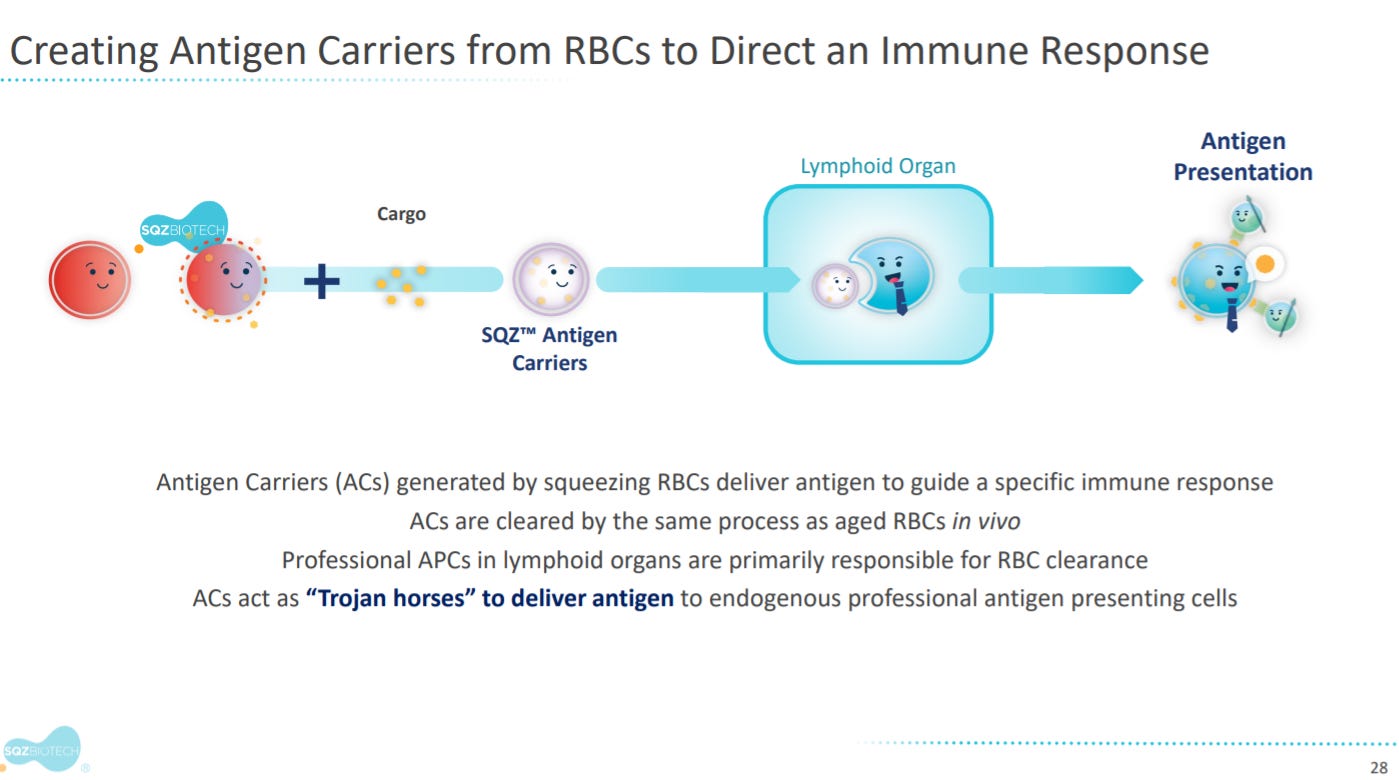
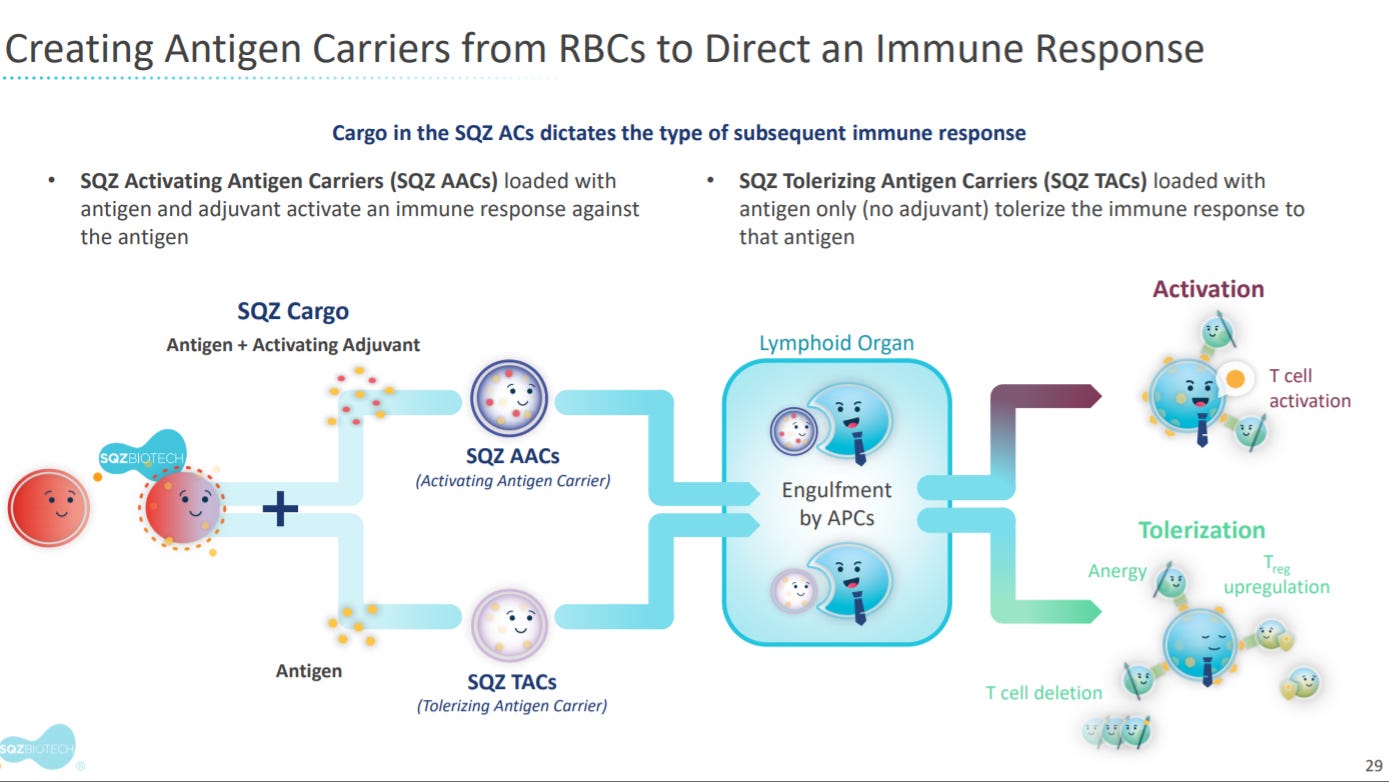
The next sections goes into SQZ’s antigen carrier products.
The premise is to engineer red blood cells (RBC) to deliver antigens to APCs in vivo.
With two mechanisms: activation and tolerization of APCs.
Section of SQZ’s Activating Antigen Carriers (AAC).
Preclinical data shows an ability to use engineered RBCs to expand CD8+ T-cells.
The company has designed a phase 1 trial of AACs in HPV-positive cancers.
Section on SQZ’s Tolerizing Antigen Carriers (TAC).
Preclinical data showed an expansion of anergic T-cells (PD1+) and Tregs.
Data in T1D models shows delay of pathology onset.
The company ends the presentation with their vision of becoming fully-integrated cell therapy company.
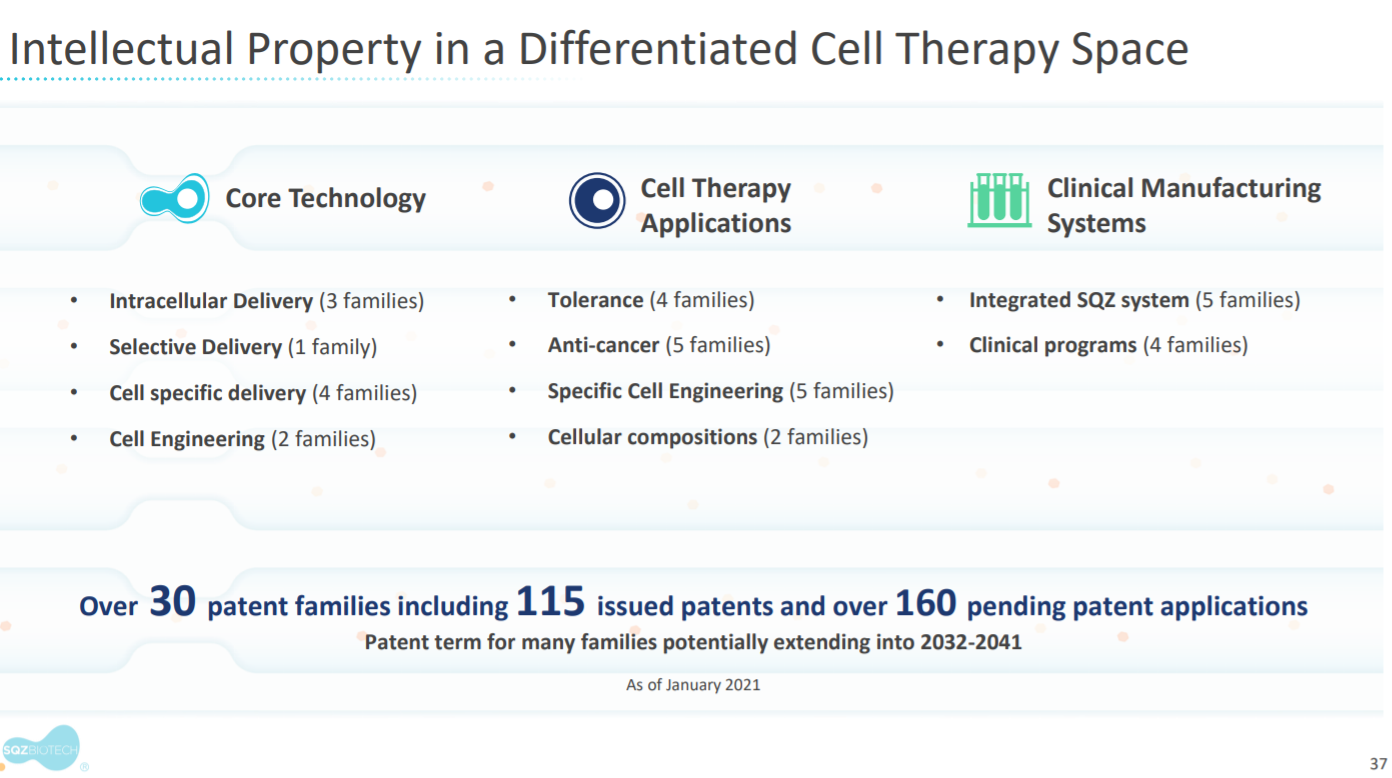
IP is a pretty important part of SQZ. In short, they can prevent other companies from squeezing cells for delivery.
The milestones is shown again.
The presentation ends with the 5-year vision.
SQZ’s presentation does a good job at showing how unique their technology platform is and their vision to scale up cell therapy manufacturing. What’s pretty profound about the company is their focus to build a pipeline of medicines rather than sell their chips similar to MaxCyte.
Follow up questions for the team:
What are the COGS and turnaround time for SQZ’s APC versus CAR-T cell therapies?
At what clinical stage does SQZ hand off their program to Roche in the partnership?
Is SQZ working to scale up manufacturing partnerships beyond Roche, AskBio, and others?