Ultralow Background Membrane Editors for Spatiotemporal Control of Phosphatidic Acid Metabolism and Signaling
Inventors & their inventions
Axial: https://linktr.ee/axialxyz
Axial partners with great founders and inventors. We invest in early-stage life sciences companies such as Appia Bio, Seranova Bio, Delix Therapeutics, Simcha Therapeutics, among others often when they are no more than an idea. We are fanatical about helping the rare inventor who is compelled to build their own enduring business. If you or someone you know has a great idea or company in life sciences, Axial would be excited to get to know you and possibly invest in your vision and company. We are excited to be in business with you — email us at info@axialvc.com
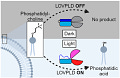
The paper describes the development of a new optogenetic tool called LOVPLD to control the production of the signaling lipid phosphatidic acid (PA) in cells with high spatiotemporal precision. PA is involved in many cellular processes but its pleiotropic nature makes its specific functions difficult to study. Previous optogenetic approaches to manipulate PA used light-induced dimerization to recruit enzymes to membranes, but had high background activity.
LOVPLD uses a different strategy by inserting a light-sensitive LOV domain into a potent PA-producing enzyme PLD to directly control its catalytic activity with light. In the dark, LOVPLD is inactive but blue light illumination causes a conformational change in the LOV domain that activates the enzyme. LOVPLD showed 50-fold lower background than previous optoPLDs and its expression was well-tolerated unlike constitutively active PLDs. Combining LOVPLD with light-induced dimerization further lowered background.
The authors first optimized the design of LOVPLD by screening different insertion sites. One version called LOVPLD P showed the best dynamic range. Experiments confirmed LOVPLD produces PA selectively in a light-dependent manner without affecting other lipids. It was localized to various organelles using targeting tags. Lipidomics after activating organelle-targeted LOVPLDs revealed differences in PA metabolism - LOVPLD activation on the ER and Golgi led to less PA accumulation than on mitochondria, PM and lysosomes, suggesting faster PA turnover on the former.
LOVPLD was then applied to study PA-mediated AMPK signaling. PA can recruit the LKB1 kinase to membranes to activate AMPK but the specific membrane sites were unclear. Imaging showed LOVPLD-generated PA recruited LKB1 to mitochondria, ER, lysosomes and Golgi but not the PM. Unexpectedly, increased AMPK activity was seen at different locations than PA production, suggesting inter-organelle crosstalk.
In summary, this paper introduced LOVPLD as a new optogenetic tool to acutely control PA production for studying its signaling roles. The protein engineering strategy of directly controlling enzyme activity via an inserted photoswitch achieved lower background than previous localization-based methods. LOVPLD enabled probing organelle-specific PA metabolism and AMPK regulation. The modular design allows combining LOVPLD with other systems for greater precision. This offers a broadly useful approach to develop optogenetic regulators of enzymes and metabolic pathways.