Build with Axial: https://axial22.axialvc.com/
Axial partners with great founders and inventors. We invest in early-stage life sciences companies such as Appia Bio, Seranova Bio, Delix Therapeutics, Simcha Therapeutics, among others often when they are no more than an idea. We are fanatical about helping the rare inventor who is compelled to build their own enduring business. If you or someone you know has a great idea or company in life sciences, Axial would be excited to get to know you and possibly invest in your vision and company . We are excited to be in business with you - email us at info@axialvc.com
Who leads Umoja Biopharma?
Umoja is one of the most unique cell therapy companies in the world right now. Pioneering in vivo cell therapies, the company has the potential to expand the number of patients who will respond to these potentially curative medicines. Founded in 2019 by Michael Jensen, Ryan Crisman (he also runs a really cool brewery in Colorado), Philip Low, and Andy Scharenberg, Umoja took IP from Seattle Children's Research Institute and Purdue University to reprogram certain immune cell populations in a given patient and redirect them against a specific cancer.
What does Umoja Biopharma do?
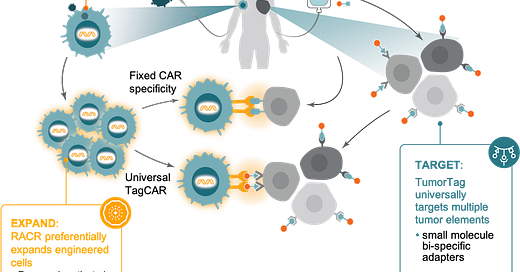
The company’s platform is built on 3 pillars:
VivoVec - the lentiviral system for delivery to endogenous T-cells. The key part is engineering the virus to resist serum inactivation and be cell-type specific. Single-chain antibodies (scFv) can be added to lentiviruses to pursue a specific target (CD8, CD20) and guide transgene delivery to T-cells. Ex vivo delivery allows for sorting of T-cells, while in vivo delivery requires a much higher bar for specificity.
RACR/CAR - standing for rapamycin activated cytokine receptor and chimeric antigen receptor respectively. This is the cargo delivered by VivoVec to expand certain cell types with the goal of avoiding patient pre-conditioning and engineer a T-cell to go after a specific target. The premise is to rely on rapamycin treatment (which is safe in humans with the right dosing) to stimulate expansion of transformed T-cells while limiting the growth of malignant cells through mTOR inhibition and most importantly removing the requirement of pre-conditioning/lymphodepletion in patients.
TumorTag - tagging cancer cells as a target for the CAR construct, via small molecules. Their first tag goes after folate receptor alpha.
“Our approach is scalable and flexible - we aspire to create immunotherapies that are accessible and valuable for patients across the full range of solid tumors.”
— Andy Scharenberg, CEO of Umoja Biopharma
By integrating 3 complementary technologies (lentivirus + protein engineering + tagging), Umoja has the potential to stimulate specific populations of immune cells and redirect them to tagged targets while avoiding non-tagged tissue as much as possible. For their business model, there are opportunities to scale and partner each of the three pillars and various combinations. Narrow deals for autoimmunity can be partnered off, the company can scale their lentiviral manufacturing capabilities, and grow their catalog of tags. Excitedly, initial data from Umoja’s development programs have been published to the public already. Umoja also recently started a phase 1 trial for an ex vivo program using the TumorTag platform.
What makes Umoja Biopharma unique?
There are a few interesting, potential, advantages of in vivo cell therapies:
The RACR/CAR technology gives Umoja the ability to sidestep preconditioning as a requirement for their therapies. On a side note, this is the value prop of B-cell therapies in the early-stages of development. Before CAR-T therapies, currently approved, a patient is given a chemotherapy regimen to clear out their T-cell repertoire (i.e. lymphodepletion) and make room for the transplanted CAR-T cells. This leads to higher levels of toxicity and adds another layer of complexity to the entire process.
In vivo cell therapies could potentially avoid the complex and costly biomanufacturing needed for cell therapies. Rather than making these medicines in an expensive facility, a patient’s own immune system produces the therapy.
Developing cancer drugs that can account for the spectrum of variants. Many candidates in oncology have unpredictable efficacy/toxicity profiles due to a high degree of cancer evolution and a suppressive tumor microenvironment, particularly for solid tumors. To expand the application of cell therapies beyond blood cancers, Umoja’s TumorTag technology could potentially help the company overcome past challenges with tumor antigen heterogeneity.
Umoja’s programs are a natural next step in the cell therapy field. The first approvals are autologous drugs (i.e. cells engineered from and given to one patient). The next programs are allogeneic or off-the-shelf - NK cell programs, led by Appia Bio, have the most promise here given their ability to penetrate solid tumors and lower rates of graft-versus-host disease (GvHD). The third act is in vivo. There are many challenges to integrate 3 technologies to make 1 drug. Umoja is validating their TumorTag platform in the clinic first with an ex vivo program as a first step. And the preclinical data looks exciting. An ability to expand certain populations of CAR-T cells and significantly reduce tumor volumes in mouse models. A lot of work is left to do: better T-cell targeting to give their in vivo programs the best chance for success, new tags for different tumors, and an ability to manufacture all of this (i.e. the process is the product).
Why I like what Umoja Biopharma is doing?
Umoja is building the next-generation of cell therapies. There are countless scientific risks to in vivo medicines. However, they offer a more scalable way to bring these complex and curative medicines to patients. Over the years several companies in the field have emerged; however, Umoja has a few underappreciated advantages that could become substantial moats over time. First, the company is investing substantially in their biomanufacturing facility in Colorado. This is due in large part from Ryan Crisman, who led the Gates Biomanufacturing Facility at University of Colorado Anschutz. Then the tumor tagging ability is a massive differentiator from others. Most platforms are focused on delivery and expansion but you will still face the issues all other cancer drugs run into - a large spectrum of variants. TumorTag imbues Umoja with the power to maximize the value of VivoVec and RACR/CAR by selective targeting of their engineered T-cells to certain tumors. But more data is needed here. Lastly, Umoja is building a diversified pipeline of in vivo and ex vivo therapies and both autologous and allogeneic/iPSC-derived. This is allowing the company to validate each of their 3 pillars in a stepwise fashion in the clinic. TumorTag with an autologous, ex vivo program. Then using RACR/CAR and VivoVec to target CD19 T-cells with an initial focus on hematological cancers. Then finally bringing all 3 technologies together to develop in vivo cell therapies for solid tumors.
You can find Umoja Biopharma here.