Immune cell decoration
Call for talented individuals and teams
Axial: https://linktr.ee/axialxyz
Axial partners with great founders and inventors. We invest in early-stage life sciences companies such as Appia Bio, Seranova Bio, Delix Therapeutics, Simcha Therapeutics, among others often when they are no more than an idea. We are fanatical about helping the rare inventor who is compelled to build their own enduring business. If you or someone you know has a great idea or company in life sciences, Axial would be excited to get to know you and possibly invest in your vision and company. We are excited to be in business with you — email us at info@axialvc.com
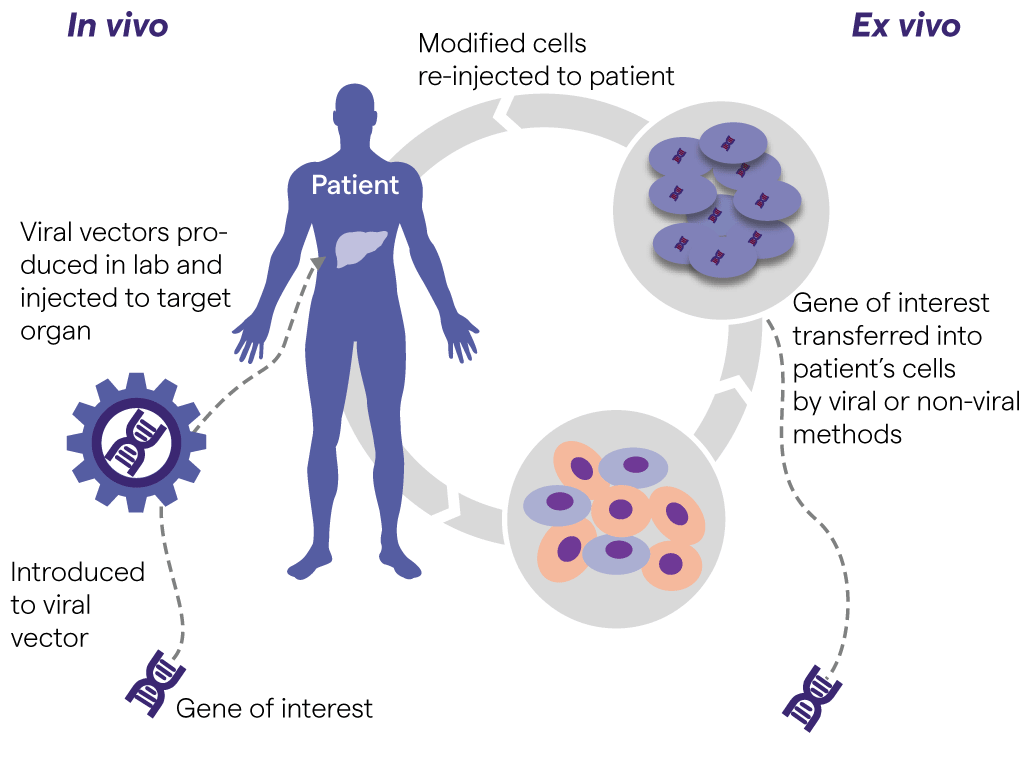
In vivo cell therapies offer immense potential to transform medicine by genetically modifying cells directly within the human body. This approach could overcome limitations of current ex vivo cell therapies like CAR-T, widen their applicability beyond blood cancers, and provide treatments for an array of genetic disorders. Companies are pursuing diverse strategies to engineer immune cells in vivo, with viral vectors, lipid nanoparticles, and other novel delivery systems to carry genetic payloads.
Decorating delivery vehicles by conjugating antibodies or other targeting moieties shows promise to guide payloads to intended cell types. For example, anti-CD3 or anti-CD8 antibodies can steer viral vectors or lipid nanoparticles towards T cells. This immune cell tropism helps concentrate the effects of gene therapies while minimizing off-target effects. Researchers have already demonstrated preclinical proof-of-concept, engineering CAR-T cells in vivo to attack tumors in mouse models of cancer.
By evading complex, personalized manufacturing, in vivo therapies may slash treatment costs and delays. Patients could receive ready-to-use infusions at local hospitals without traveling to specialized centers. The immediacy could help patients with aggressive, fast-moving cancers that worsen during prolonged CAR-T production. Avoiding pre-infusion chemotherapy conditioning regimens further benefits patients.
In vivo approaches do pose challenges. Precise targeting is paramount to avoid potentially dangerous off-target effects, like inadvertently modifying cells that trigger graft-vs-host disease. Developers must balance transient expression to minimize safety risks with integration into genomes for sustained, durable benefits. And while lipid nanoparticles avoid genomic changes, their effects are temporary as cells dilute out the genetic instructions.
Viral vectors can integrate genetic payloads to provide lasting traits, but even gutted, non-replicating viruses may trigger unintended immune responses. Lentiviruses also carry some cancer mutation risks that'll require careful toxicology assessments. Regulators will scrutinize specificity of delivery vehicles and monitor for secondary malignancies.
Some companies like Sana Biotechnology are focused on hereditary blood disorders, aiming to correct disease-causing mutations in hematopoietic stem cells. Editing stem cells could cure sickle cell disease, beta thalassemia and other genetic illnesses. Other groups target different cell types, hoping to treat heart disease, kidney fibrosis or neurodegeneration by providing cells with instructions to make protective proteins.
Beyond immunotherapy, in vivo gene editing of stem cells may transform treatment for thousands of inherited disorders. But delivering gene editors like CRISPR/Cas9 to the right tissues and cells in vivo remains challenging. Innovations like using adeno-associated viruses as delivery shuttles are active areas of research.
While recent entrants aim for broad applicability across many disease areas, realizing that full potential will take years. Most existing work remains preclinical. The few companies with named drug candidates expect to start human testing within the next 1-2 years. These early clinical trials will focus on safety and feasibility. Should results warrant, developers will then have to validate efficacy through lengthy, rigorous studies.
In vivo cell engineering may democratize access to emerging genetic medicines, but costs won't disappear overnight. Though likely cheaper than current ex vivo cell therapies, in vivo treatments will still carry price tags reaching hundreds of thousands initially. The viral vectors and nanoparticles required are complex to manufacture, and will take time to scale production.
Some experts are also concerned resistant cancers could emerge, just as they have for CAR-T. Target cells could evolve to dodge therapies by dropping or modifying proteins tumors use to evade detection. Combinations with traditional cancer drugs could help prevent resistance.
Despite outstanding questions, the transformative premise of generating “living drugs” inside the human body has captured imaginations. Visionary scientists see potential to erase the underlying genetic culprits behind cancers and genetic diseases. Skilled execution by talented teams at emerging companies now needs to translate these visions into safe, effective new medicines for patients worldwide.