i-shaped antibody engineering enables conformational tuning of biotherapeutic receptor agonists
Inventors & their inventions
Axial: https://linktr.ee/axialxyz
Axial partners with great founders and inventors. We invest in early-stage life sciences companies such as Appia Bio, Seranova Bio, Delix Therapeutics, Simcha Therapeutics, among others often when they are no more than an idea. We are fanatical about helping the rare inventor who is compelled to build their own enduring business. If you or someone you know has a great idea or company in life sciences, Axial would be excited to get to know you and possibly invest in your vision and company. We are excited to be in business with you — email us at info@axialvc.com
The paper describes a new approach to engineer antibodies to act as agonists for cell surface receptors, which has potential applications in developing new biotherapeutics. The authors developed a method to re-shape conventional Y-shaped antibodies into more compact, linear "i-shaped" antibodies (iAbs) by transplanting sets of mutations identified in naturally occurring broadly neutralizing antibodies against HIV. Adding a new approach to the antibody engineering tool box - iAb engineering.
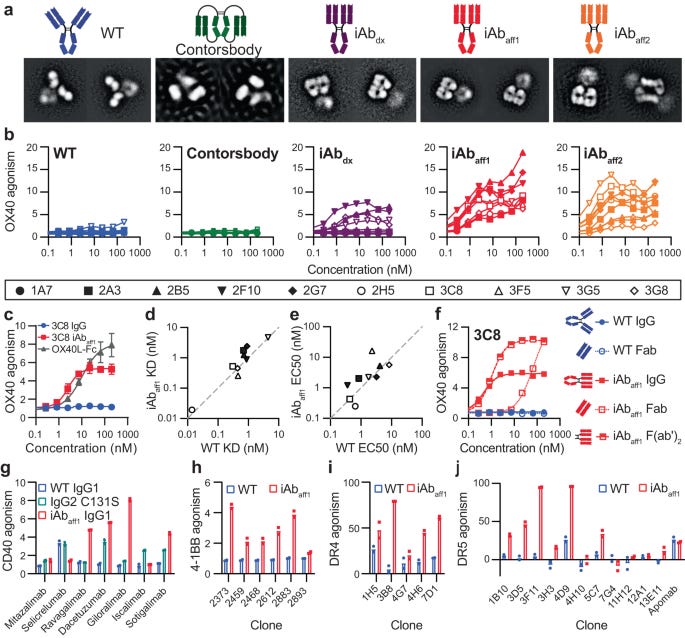
Three sets of mutations termed iAbdx, iAbaff1, and iAbaff2 were engrafted into panels of 10 antibodies targeting OX40 and induced the iAb shape, as confirmed by negative stain electron microscopy. iAbaff1 and iAbaff2 were most effective at inducing the iAb conformation. All iAb variants of the anti-OX40 antibodies gained intrinsic OX40 agonism activity in a cell-based reporter assay, while the unmodified Y-shaped antibodies had no activity. The iAbaff1 mutations provided the strongest and most consistent agonism. The enhanced activity was not due to differences in antigen binding affinity or avidity compared to unmodified antibodies, suggesting the iAb shape itself enables receptor agonism. TIRF microscopy indicated the iAb format causes local OX40 clustering.
Antibody panels targeting other TNFRSF members (CD40, 4-1BB, DR4, DR5) also gained intrinsic agonism when converted to the iAb shape using the iAbaff1 mutations. This underscores the broad utility of this approach. A bispecific iAb format was applied to activate the heterodimeric IL-2 receptor. Various anti-IL2Rβ and anti-IL2Rγ antibodies were screened in bispecific IgG and iAb combinations. While unmodified IgGs had no activity, some iAb variants demonstrated IL-2 mimetic activity. Lead iAb agonists induced dose-dependent proliferation of primary NK and T cells akin to native IL-2. Transcriptomic profiling also indicated the iAbs induce an IL-2-like gene signature.
In comparison to multivalent IgG hexamers, the bivalent iAb format provided strong OX40 agonism and receptor clustering with more moderate effects on receptor internalization and confinement. This supports mechanistic differences between multivalent versus conformation-based strategies. Overall, the iAb approach enables potent antibody agonism against diverse receptors by conferring a unique mechanism of action. The conventional IgG-like nature offers potential advantages as a therapeutic modality compared to other non-native formats. While promising, open questions remain regarding translational potential, effects on developability, understanding specificities in mechanisms of action, and determining rules guiding applications to new receptors. Investigating these areas is needed to advance iAbs toward the clinic.
In summary, this paper introduces iAb engineering as a promising technology to create antibody agonists by modulating antibody shape. A new approach for agonist antibody engineering, inspired by nature, that circumvents the need for extrinsic factors to drive potent agonism. Applied to TNFR superfamily members and the heterodimeric IL-2R. The broad success against receptors from two different classes highlights the potential generalizability. This approach could enable new antibody-based drugs that mimic beneficial biological signals for treating disease. If the remaining open questions and risks can be addressed through further research, iAbs may eventually provide clinical benefit for patients.