Axial: https://linktr.ee/axialxyz
Axial partners with great founders and inventors. We invest in early-stage life sciences companies such as Appia Bio, Seranova Bio, Delix Therapeutics, Simcha Therapeutics, among others often when they are no more than an idea. We are fanatical about helping the rare inventor who is compelled to build their own enduring business. If you or someone you know has a great idea or company in life sciences, Axial would be excited to get to know you and possibly invest in your vision and company. We are excited to be in business with you — email us at info@axialvc.com
How we can reverse epigenetic damage at the cellular level?
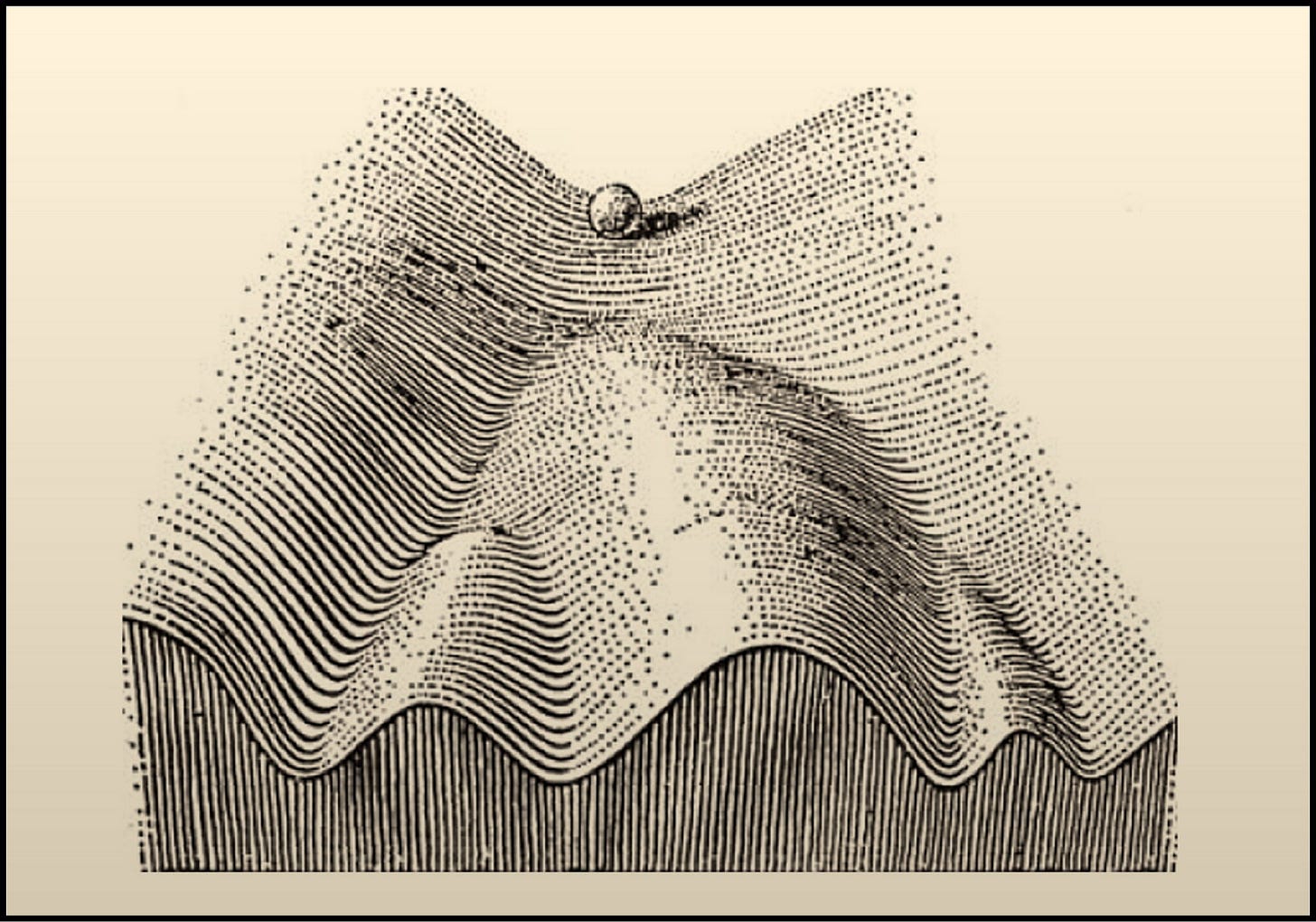
The epigenetic theory of aging posits that aging is driven primarily by the loss of epigenetic information in cells over time. This loss causes cells to essentially "forget" their identity and proper function, leading to the progressive decline and dysfunction we observe in the aging process. Which implies that by restoring this lost epigenetic information could effectively reset aged cells back to a more youthful, properly functioning state.
The idea that aging has an epigenetic basis is supported by several key lines of evidence. First, the work of John Gurdon in the 1960s demonstrated that the nucleus from an aged somatic cell, when transplanted into an enucleated egg cell, could give rise to a healthy young organism. This implied that the aging process must involve some form of reversible epigenetic changes, rather than being purely encoded in the genome sequence itself.
Building on this, Shinya Yamanaka's pioneering work a few decades later showed that introducing just four transcription factors could reprogram differentiated somatic cells back into an induced pluripotent stem cell (iPSC) state that was molecularly and functionally indistinguishable from an embryonic stem cell. Crucially, cells reprogrammed into iPSCs displayed a complete "reset" of aging hallmarks like telomere attrition, mitochondrial dysfunction, and gene expression changes - they became molecularly youthful once again.
Subsequent research has confirmed that the reprogramming process leading to iPSCs effectively wipes cells' epigenetic slate clean, reverting their epigenomes to an embryonic-like ground state. This resetting of the epigenome appears both necessary and sufficient for conferring age reversal of cellular traits. Methods like direct transdifferentiation that change a cell's identity without passing through a pluripotent intermediate do not lead to the same age reversal effects.
The ability to reverse cellular aging via epigenetic reprogramming has now been demonstrated across multiple cell types, including fibroblasts, neurons, and others. Aged cells reprogrammed into iPSCs and then re-differentiated into the same lineage appear biochemically and functionally rejuvenated, indistinguishable from counterparts derived from young donors.
So the basic premise that aging is driven by epigenetic drift, and that this epigenetic dysregulation can be reversed by resetting the epigenome, seems well-supported by the available evidence from cellular reprogramming studies. Aging certainly involves other factors like DNA mutations, but the epigenetic component appears to be a key root cause that can be targeted.
The bigger challenge is translating this knowledge into therapies that could rejuvenate aging tissues and organs in living organisms, not just cultured cells. Simply introducing Yamanaka's reprogramming factors throughout the body could lead to teratoma formation from any cells that get fully reprogrammed into an uncontrolled pluripotent state.
Here the idea of "partial reprogramming" using intermittent, restricted dosing of reprogramming factors aims to guide cells part of the way to the pluripotent state, resetting their epigenomes enough to remove age-associated epigenetic noise while avoiding complete dedifferentiation. Early studies show this can extend lifespan and healthspan in progeroid and wildtype mice, seemingly through cellular rejuvenation mechanisms.
However, implementing partial reprogramming will require extremely tight control to ensure cells get reprogrammed just the "right" amount and don't pass the point of no return into a full pluripotent state. We currently lack the sophisticated gene circuit tools that would enable such exquisite spatiotemporal regulation of the reprogramming process in every cell.
Beyond just reprogramming individual cells, there are also broader questions around whether epigenetic rejuvenation alone will suffice to comprehensively reverse aging in entire tissues and organisms. Aging impacts higher-order tissue architecture and cell ratios in ways that may not be reversible by de-aging residual cells. Some aging pathologies like complete tooth or limb loss almost certainly require regenerative interventions beyond just cellular reprogramming.
So while the epigenetic theory convincingly explains cellular aging and provides provocative examples of age reversal in cultured cells, extending this to whole-organism rejuvenation faces major hurdles. Developing sufficiently precise reprogramming control methodologies and understanding whether reprogramming can fully restore aged tissue systems are key challenges ahead. Nonetheless, the tantalizing prospect of epigenetic age reversal has understandably captivated the imagination of researchers, funders, and the broader public. If the epigenetic theory holds true, it could finally yield a general solution to the "unsolved problem" of biological aging that has vexed humanity for centuries.
For the first time, we may be glimpsing the existence of a biologically encoded rejuvenation program hard-wired into our cells - one that evolution inserted for the purely selfish purpose of restarting the life cycle in each generation via reproduction. By hijacking and amplifying these same molecular pathways, therapies based on epigenetic reprogramming could effectively keep "rebooting" our cells to maintain them in a perpetual state of youth. Of course, the full scope of aging's root causes likely extends beyond just epigenetic drift to include contributions from molecular damage, loss of regenerative capacity, metabolic dysregulation, and other factors. Interventions like senolytics to remove dysfunctional senescent cells accumulating with age may prove synergistic when combined with epigenetic reprogramming approaches.
But by potentially solving a key driver of aging - the gradual erosion of cellular identity and function encoded in the epigenome - the epigenetic theory provides one of the first unifying explanations for how and why we age at a fundamental level. It exposes aging not as a static, irreversible process but as one of dynamic dysregulation that could potentially be corrected by reinstating cells' proper epigenetic instruction manuals.
The concept of aging as simply a loss of biologically encoded information, one that our cells already possess the latent capacity to restore, represents a profound paradigm shift in our understanding of senescence. It reduces a seemingly intractable, inevitably entropic process to an engineering challenge of maintaining proper data integrity and storage - a "Software 1.0" problem that our increasingly sophisticated biotechnologies may allow us to circumvent.
Just as we can reinstall corrupted software on our computers to restore full functionality, the epigenetic theory suggests we may one day be able to reflash our very cells and bodies, reverting age-associated declines by refreshing their internal epigenetic code. It's the biological equivalent of the most sought-after innovation in human history - a way to wind back the clock of life itself. Realizing this transformative vision in a safe, controlled, and equitable way poses immense scientific and ethical challenges that we are only beginning to grapple with. But the epigenetic theory shows why the relentless march of biological aging may not be as inevitable as it once seemed. If its conceptual premises prove correct, it could finally pan out into the long-mythologized fountain of youth - a way to eternally renew ourselves at life's ultimate source code layer.