Engineering immune tolerance
Inventors & their inventions
Axial: https://linktr.ee/axialxyz
Axial partners with great founders and inventors. We invest in early-stage life sciences companies such as Appia Bio, Seranova Bio, Delix Therapeutics, Simcha Therapeutics, among others often when they are no more than an idea. We are fanatical about helping the rare inventor who is compelled to build their own enduring business. If you or someone you know has a great idea or company in life sciences, Axial would be excited to get to know you and possibly invest in your vision and company. We are excited to be in business with you — email us at info@axialvc.com
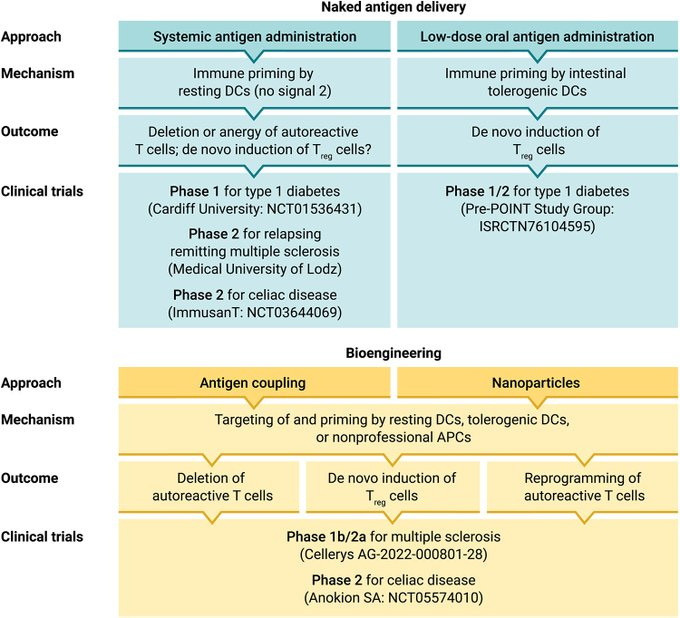
Autoimmune disorders encompass a wide range of immunological diseases that affect 4%-10% of the population worldwide. They occur when immunological tolerance toward self-antigens is broken, resulting in immune responses against cells, tissues, or organs that lead to tissue dysfunction and/or destruction. The goal of autoimmune disease therapy is to restore tolerance to the self-antigen that causes the pathology by targeting autoreactive T-cells while preserving immune competence to prevent infections and malignancies. The main challenge of this endeavor resides in choosing the mode by which the disease-driving antigen is delivered so that it can initiate the removal or reprogramming of the autoreactive T-cells or the induction and/or expansion of antigen-specific regulatory T (Treg) cells to suppress autoreactive T cells. Although the potential of antigen-based immunotherapy approaches to restore tolerance have been demonstrated for the treatment of immunoglobulin E (IgE) – mediated peanut allergy, antigen therapies for autoimmune disorders are still being developed.
Several antigen delivery strategies are being explored to reverse autoimmunity. One approach is to administer the antigen in its native form to induce T cell deletion or anergy. However, this carries the risk of inducing inflammation and exacerbating disease. To avoid this, the antigen dose and route of administration must be carefully controlled. Low doses of antigen administered orally or through the skin promote tolerance through deletion or anergy of autoreactive T cells and expansion of Tregs. Higher antigen doses or systemic administration are more likely to activate autoreactive T cells. Another consideration is whether to use the whole antigen or just critical T cell epitopes. The advantage of using whole antigens is that all potential epitopes are targeted. However, dominant epitopes may provoke inflammation. Using select immunodominant epitopes may allow tolerance to be induced more safely.
Encapsulating the antigen in nanoparticles is another promising approach to modulate antigen delivery. Nanoparticles protect antigens from degradation, facilitate uptake by antigen presenting cells (APCs), and allow for controlled antigen release. They also reduce systemic exposure. Various biodegradable nanoparticles made of polymers, lipids, or albumin have been studied. Nanoparticles can be designed to target APCs through surface modifications. Targeting nanoparticles to dendritic cells in lymph nodes allows antigens to be delivered directly to critical sites of T cell activation and tolerance induction. Co-delivery of immunosuppressive drugs with antigens in nanoparticles may enhance their tolerogenic effects.
Using antigen-coupled cells is another way to deliver antigens and promote tolerance. Both immature and semi-mature dendritic cells have tolerogenic properties that can be exploited. Coupling disease-relevant antigens to these cells before infusion allows for efficient antigen presentation in a tolerogenic context. Antigen coupled apoptotic cells are also being investigated. Phagocytosis of apoptotic cells promotes tolerance through anti-inflammatory effects and expansion of Tregs. Coupling antigens to apoptotic cells harnesses this tolerogenic potential. Mesenchymal stem cells likewise have immunomodulatory properties that make them attractive antigen carriers. Genetic engineering can be used to load antigens onto cells to create "tolerogenic vaccines".
Attenuated bacterial and viral vectors provide another means to deliver antigens in a tolerogenic fashion. Listeria, Salmonella, adeno-associated virus, and lentivirus vectors have been engineered to express specific autoantigens and successfully reversed disease in animal models. Using microbial vectors has the added benefit of eliciting innate immune signals that promote tolerogenesis. However, pre-existing immunity against vectors can reduce efficacy over time. Also, there are safety concerns regarding possible reversion to virulence.
Finally, implantable biomaterial scaffolds containing antigens offer a way to achieve local, sustained antigen delivery. Scaffolds composed of biopolymers like collagen have been used to reverse autoimmunity by steadily releasing antigens and modulating inflammation in the local tissue microenvironment. Scaffolds can also be designed to attract and activate antigen-specific Tregs. Further study is needed to identify biomaterials that safely promote robust, long-lasting tolerance.
Modulating antigen delivery through careful selection of dose, route of administration, use of tolerogenic cells or vectors, and biomaterial scaffolds represents a promising therapeutic strategy to reverse autoimmunity. Key challenges moving forward include identifying the critical antigens and epitopes that drive specific autoimmune diseases, optimizing antigen delivery approaches to safely induce stable tolerance without generalized immunosuppression, and developing systems for regulated or targeted antigen delivery. Continued research in this area offers hope for antigen-specific immunotherapies that can eventually reduce the burden of autoimmune disease.