Axial partners with great founders and inventors. We invest in early-stage life sciences companies such as Appia Bio, Seranova Bio, Delix Therapeutics, Simcha Therapeutics, among others often when they are no more than an idea. We are fanatical about helping the rare inventor who is compelled to build their own enduring business. If you or someone you know has a great idea or company in life sciences, Axial would be excited to get to know you and possibly invest in your vision and company . We are excited to be in business with you - email us at info@axialvc.com
Urovant was founded in 2017 after spinning out of Roivant Sciences to develop new medicines in urology. The company’s initial business model was to initiate a multinational, registrational phase 3 trial for vibegron, which was licensed from Merck, a phase 3-ready selective β3-adrenergic agonist for the treatment of overactive bladder. At founding, over 2,700 patients with overactive bladder had been enrolled in clinical trials for vibegron: a phase 2b study for 1,300 patients meeting its primary endpoint and a phase study of 1,000 patients. Overtime, if this phase 3 trial saw success Urovant would build out a broader pipeline in urology.
The first slide of their latest corporate presentation explains their vision of becoming a fully-integrated urology drug development company and various parts of their business.
Urovant goes directly to a team slide.
Then the company explains their catalysts: PDUFA for vibegron, which was approved by the FDA in early 2020, potential sales of vibegron, and the potential of the medicine to expand into larger indications.
Urovant’s gives an overview of their pipeline: using vibegron has a pipeline with a drug with a gene therapy for overactive bladder.
After these overview slides, the company goes into the clinical data for their lead asset.
The next slide goes into Urovant’s clinical plan. β3-adrenergic agonists have been in development since the 1990s initially for obesity and type 2 diabetes because β3-adrenergic receptors (GPCR) are involved in lipolysis and thermogenesis. However, several of these trials failed. It has been known for decades that the urinary bladder in humans is innervated with both sympathetic and parasympathetic nerves with adrenergic stimulation inducing bladder relaxation. On a side note, cholinergic stimulation induces contraction. But, β-adrenergic receptors in the gut were not within the known 1/2 subtype class. In 1999, β3-adrenergic receptors were found to be the key driver for bladder relaxation - https://pubmed.ncbi.nlm.nih.gov/10188996/
This discovery set the basis for the approval of Mirabegron in OAB and the development of Urovant’s Vibegron.
Urovant presents the results from their pivotal phase 3 trial. Versus tolterodine, a SoC for OAB, Vibegron shows significant improvement across the trial’s primary and secondary endpoints.
Urovant shows Vibegron’s long-term ability to reduce daily urge urinary incontinence (UUI) episodes in OAB patients, which are urges that lead to involuntary urine leakage.
They also show Vibegron’s ability to reduce UUI episodes in patients with acute OAB.
With efficacy data shown, Urovant shows a roughly comparable safety profile of Vibegron with the standard-of-care.
Urovant uses the last 3 slides on data to argue why Vibegron is differentiated with Mirabegron from Astellas. Urovant needs to shown more data to substantiate these claims.
After giving an overview on their lead asset, Urovant goes into their commercial strategy for the medicine.
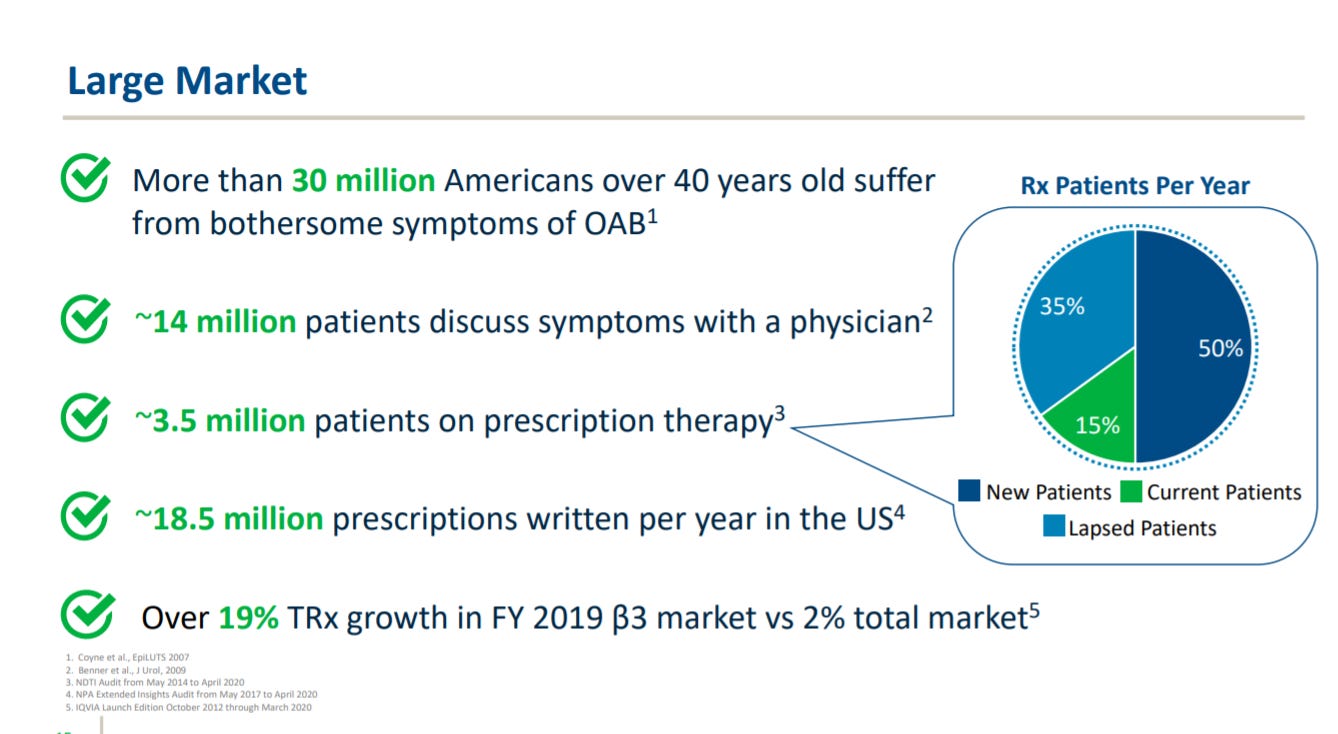
The company gives an overview of the OAB market. It’s more of a chronic disease leading to discomfort rather than mortality (although there is some.
Urovant provides their commercial milestones for Vibegron in OAB: create their own sales force and convert as many primary care physicians and urologists as possible to the medicine.
Urovant goes deeper into the sales strategy - focusing on physicians, urologists, and long-term care patients.
On the other side, Urovant is working on ensuring the payor side is figured out before launch.
Urovant is also building up their own marketing strategy to raise OAB patient awareness of Vibegron. Drug marketing especially with the Internet has a lot of room to improve. Roy Vagelos has had some of the best ideas here.
Urovant goes into their growth strategy beyond OAB.
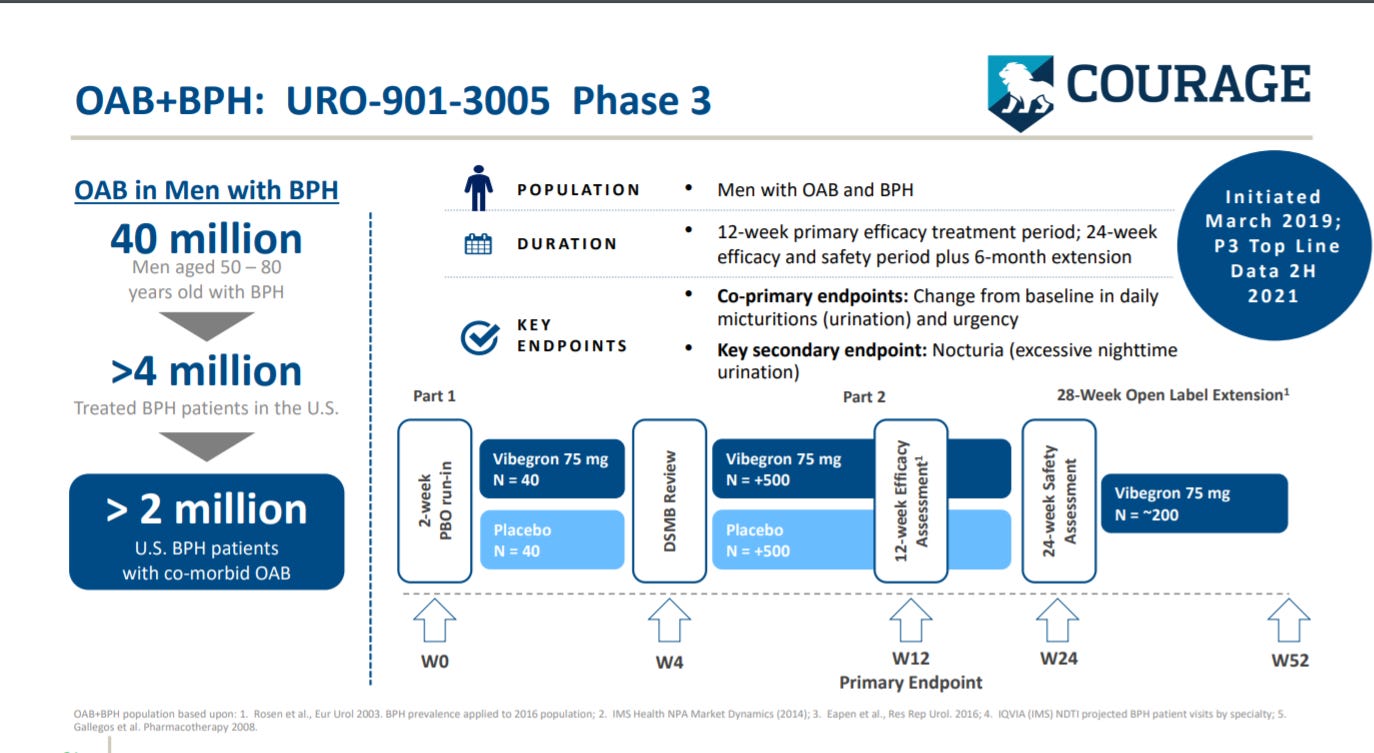
Urovant goes into their second trial of Vibegron for patients with OAB and benign prostatic hyperplasia (i.e. prostate enlargement), which is in phase 3 studies.
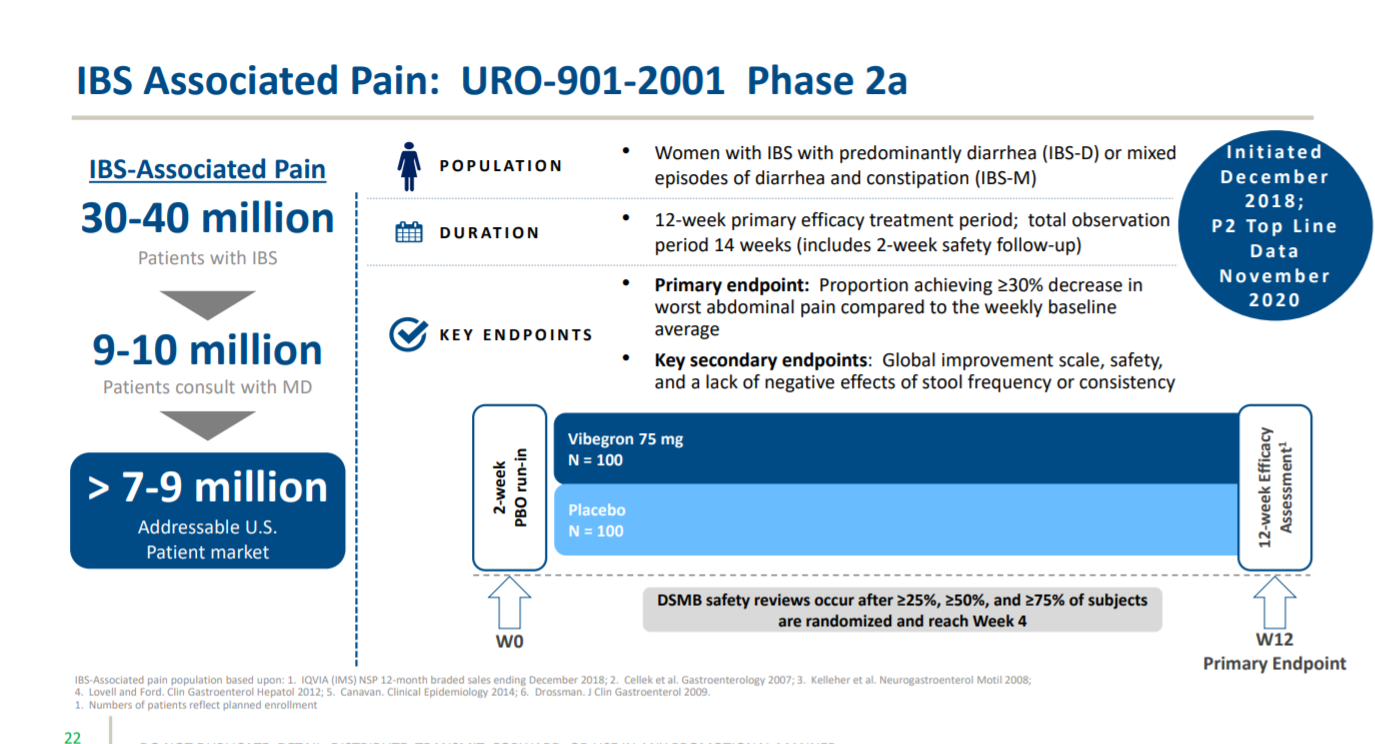
Then the third trial of Vibegron for irritable bowel syndrome associated pain, which is in phase 2 trials.
Finally, Urovant gives an overview of their gene therapy (injected directly in the gut) asset for OAB in phase 2 trials.
Urovant’s penultimate slide goes into their milestones - the ones they have achieved mainly getting Vibegron approved and their future ones, scaling up sales.
Urovant then reuses their first slide on an overview of the company.
Urovant’s deck repeatedly conveys the importance of Vibegron’s approval and does a good job at alluding to the medicine’s potential to treat multiple urological diseases.
Follow up questions for the team:
What data suggests Vibegron’s advantages over Miraberon in OAB? This will influence the success of the sales strategy.
By what logic is Urovant choosing new indications for Vibegron to pursue?