Axial partners with great founders and inventors. We invest in early-stage life sciences companies such as Appia Bio, Seranova Bio, Delix Therapeutics, Simcha Therapeutics, among others often when they are no more than an idea. We are fanatical about helping the rare inventor who is compelled to build their own enduring business. If you or someone you know has a great idea or company in life sciences, Axial would be excited to get to know you and possibly invest in your vision and company . We are excited to be in business with you - email us at info@axialvc.com
NGM Biopharmaceuticals is a business focused on large markets with diseases driven by clear mechanisms-of-action. The company was founded in 2007 by Jin-Long Chen, the company's chief scientific officer who was president up until 2014. Chen was the VP of Biology at a biotech company called Tularik that was ultimately bought by Amgen for $1.3B. Chen earned his PhD in the lab of one of Tularik's co-founders, Professor Robert Tjian. While at Amgen, Chen served as VP of Metabolic Disorders. NGM was founded on the premise of drugging metabolic and endocrine disorders with clear drivers that can be targeted with a drug.
NGM's business model is focused on 4 parts:
Is the target NGM could pursue the key driver of the disease?
Is the disease a large market?
Could the current toolkit of drug discovery and biology, successfully, generate a medicine for the target and disease?
Is there a large unmet clinical need?
This model is very focused on finding opportunity with a margin of safety - clear biology with large markets. This framework has led the company to build a de-risked pipeline in non-alcoholic steatohepatitis (NASH), which represents over $20 billion in potential revenue with no approved drugs for the disease. Liver disease and NASH are characterized by reduced levels of the human hormone fibroblast growth factor 19 (FGF19), an endocrine hormone that controls bile acid synthesis for the liver. Defective FGF19 levels are thought to lead to NASH. In order to restore liver health, NGM's Phase 2b lead program (Aldafermin, NGM282) is focused on delivering daily an engineered variant of FGF19 to patients. This internal program is the leading example of NGM's business model to find large markets with defined mechanisms and bring their team's expertise and knowledge base to bear to develop new medicines.
The first slide of their latest corporate presentation lays out the statuses of their pipeline, partnerships, and team. The first slide ideally would lay out their unique business model and mission.
The next slide gives an overview of NGM’s pipeline.
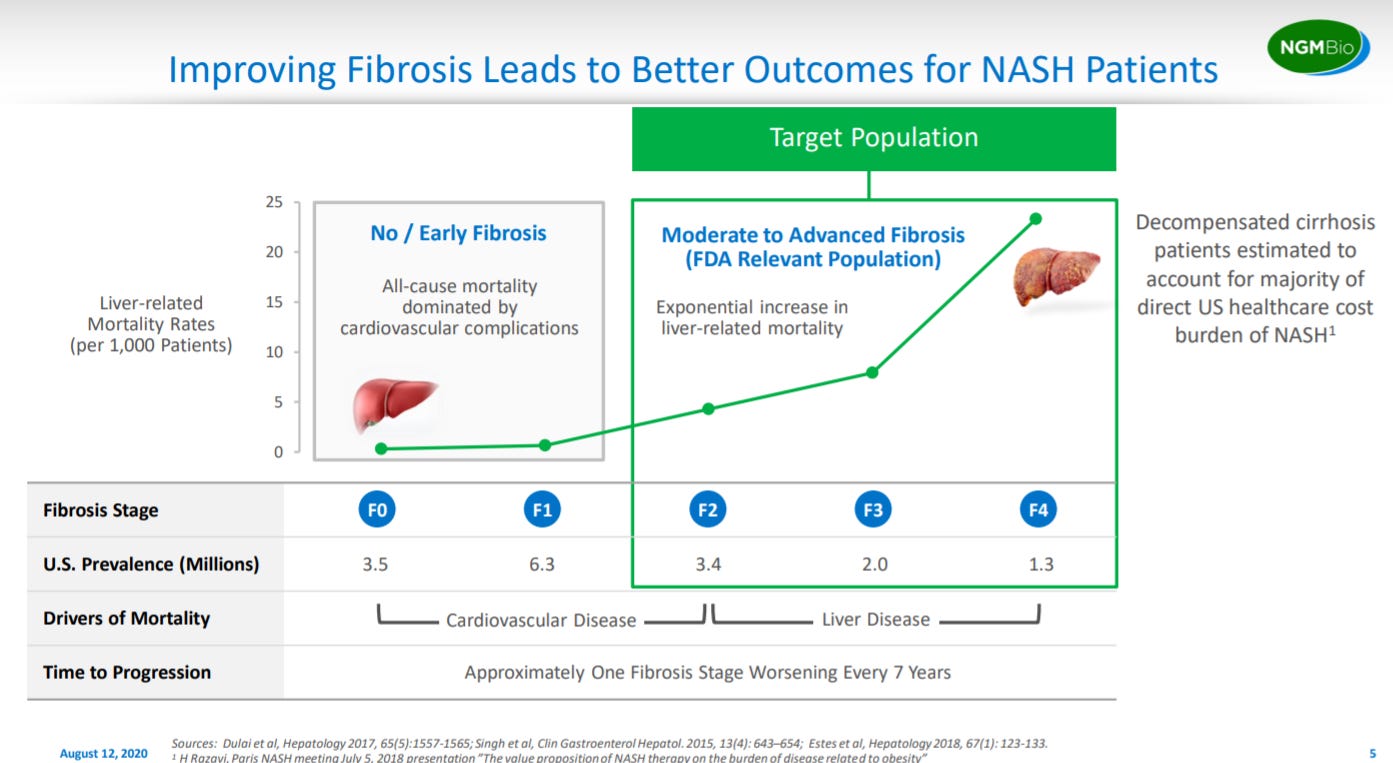
Then the company goes into their premise to treat late-stage NASH where most of the liver-related mortality is generated.
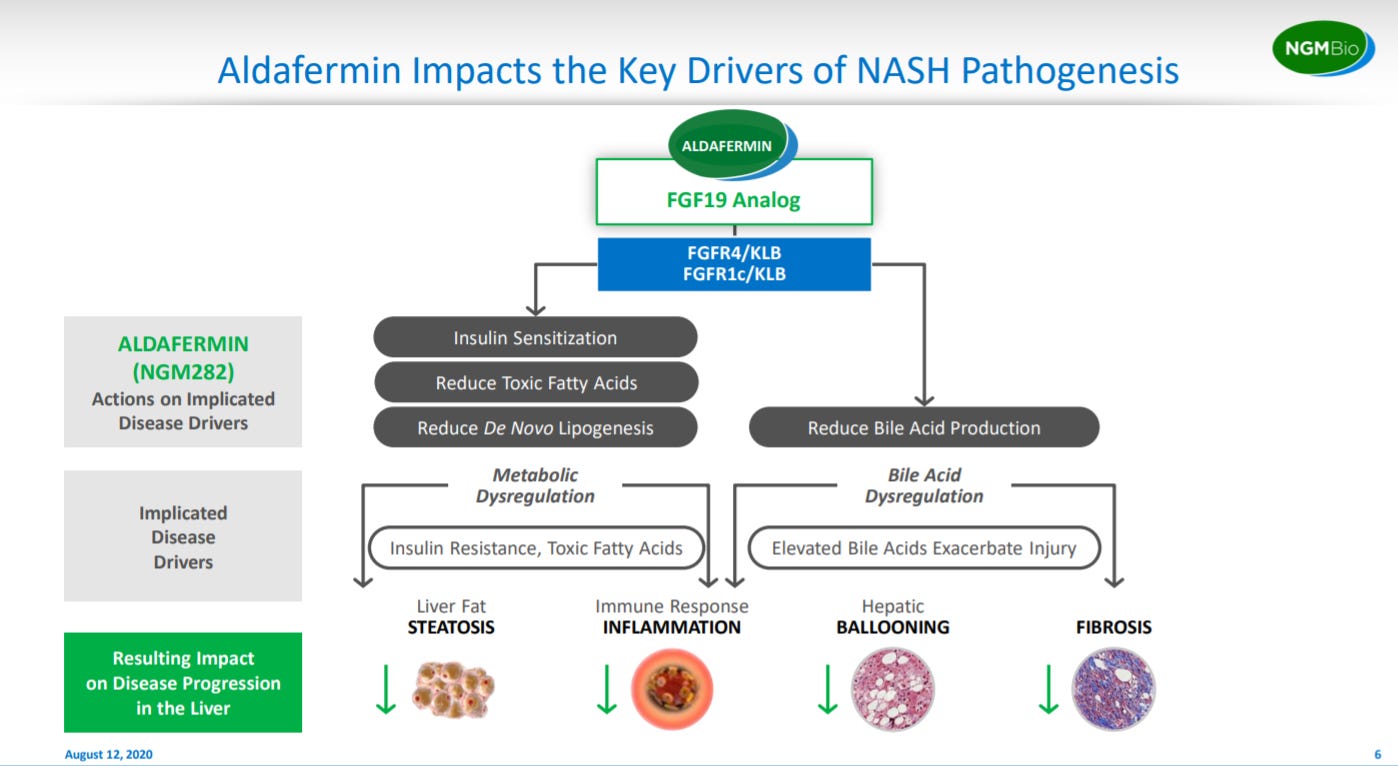
After laying out their biological hypothesis, NGM explains the MoA for their lead asset: Aldafermin.
NGM then tries to compare Aldafermin to other strategies. Mainly, they are focusing on reversing fibrosis to treat NASH instead of preventing it.
The presentation mainly focused on giving an update on the last patient cohort of their phase 2 trial for Aldafermin.
This slide explains the trial design - endpoints, inclusion criteria, duration.
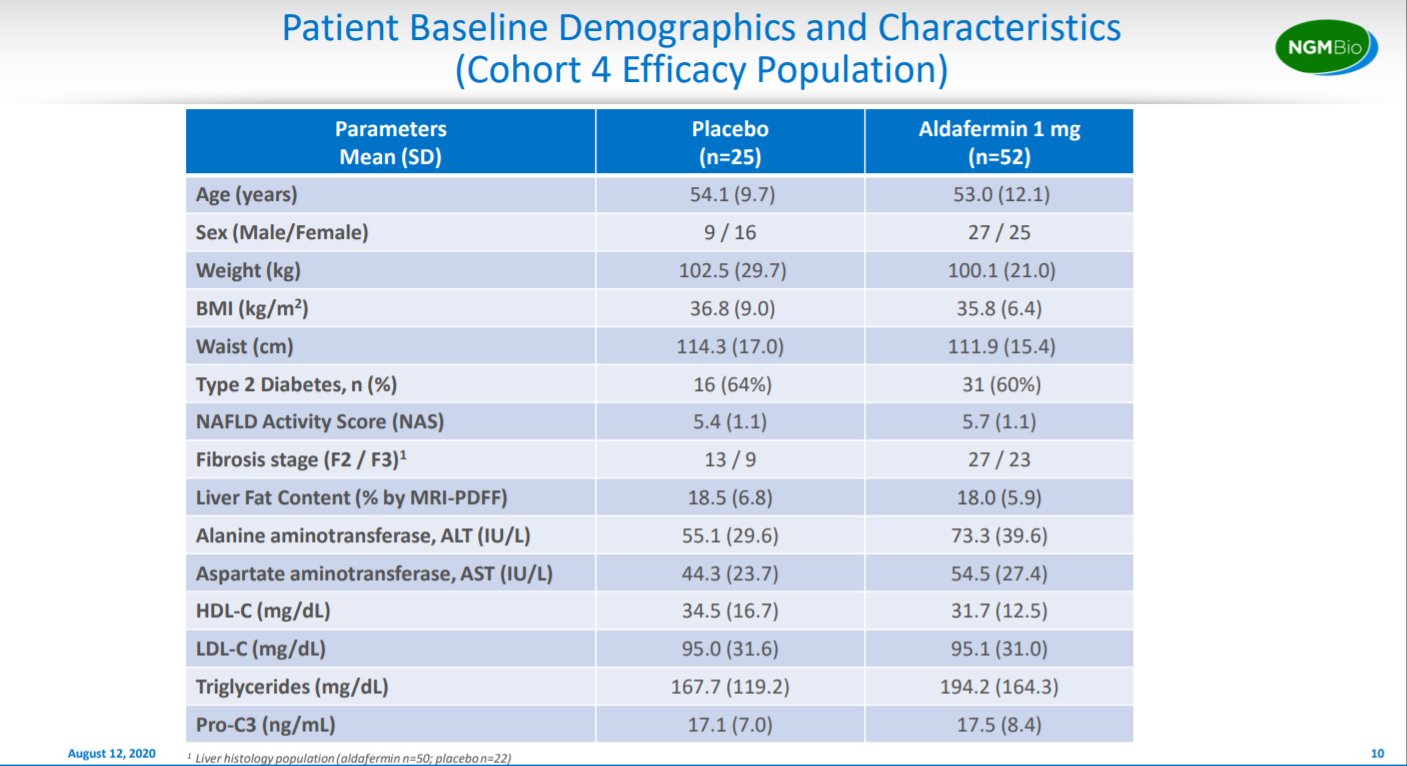
This is a pretty useful slide to compare demographics within the cohort between the placebo the treatment arms.
NGM reports that after 24 weeks, ~50% more patients show improvements in fibrosis from a liver biopsy (measuring liver fat content) versus placebo. They should clarify significance.
Twelve patients saw NASH resolution after 8 months verified by liver biopsy.
They are showing the previous two figures in comparison to results for two other cohorts with one at a different dose. It’s not clear what the point of this slide is.
They then show a significant result that 11 patients versus 0 in the placebo show both fibrosis reversal and resolution of NASH.
NGM shows significant improvement of NAS (a score for liver injury) for a large part of the treatment arm in the cohort.
This is the key slide where NGM’s drug candidate meets the primary endpoint of the trial: reduce liver fat content versus placebo and baseline.
Moreover, they show that the drug candidate meets its secondary endpoint: significantly reduce levels of ALT (an aminotransferases) shown to be elevated in NASH patients.
This slide goes into a potential sid-eeffect of Aldafermin - elevated LDL-C (i.e. bad cholesterol). This slide shows that the drug candidate does increase LDL-C initially but then goes back to normal levels for these patients.
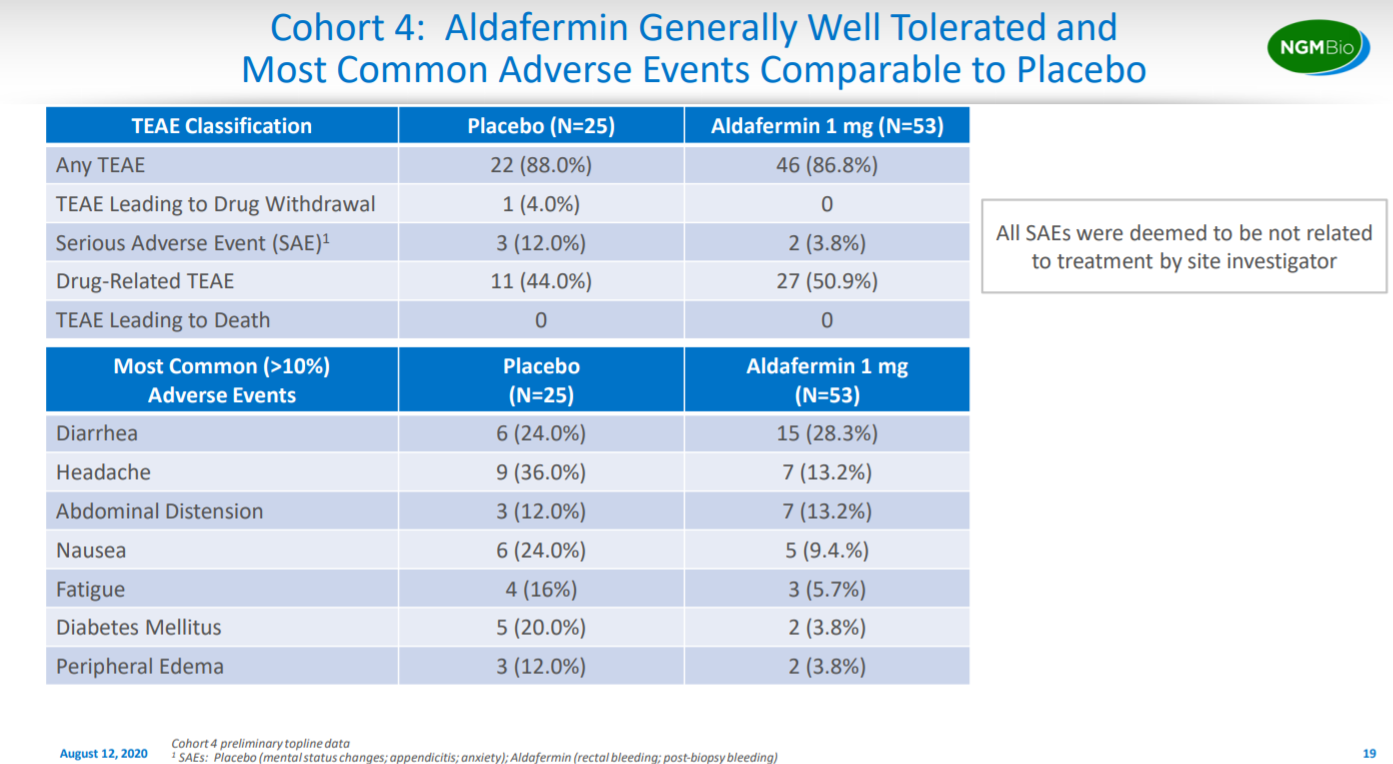
This slide goes into the safety for the cohort.
This slide just synthesizes all the results for the phase 2 data for cohort 4. Pretty exciting data.
NGM wraps it all up with a summary of the cohort 4 results.
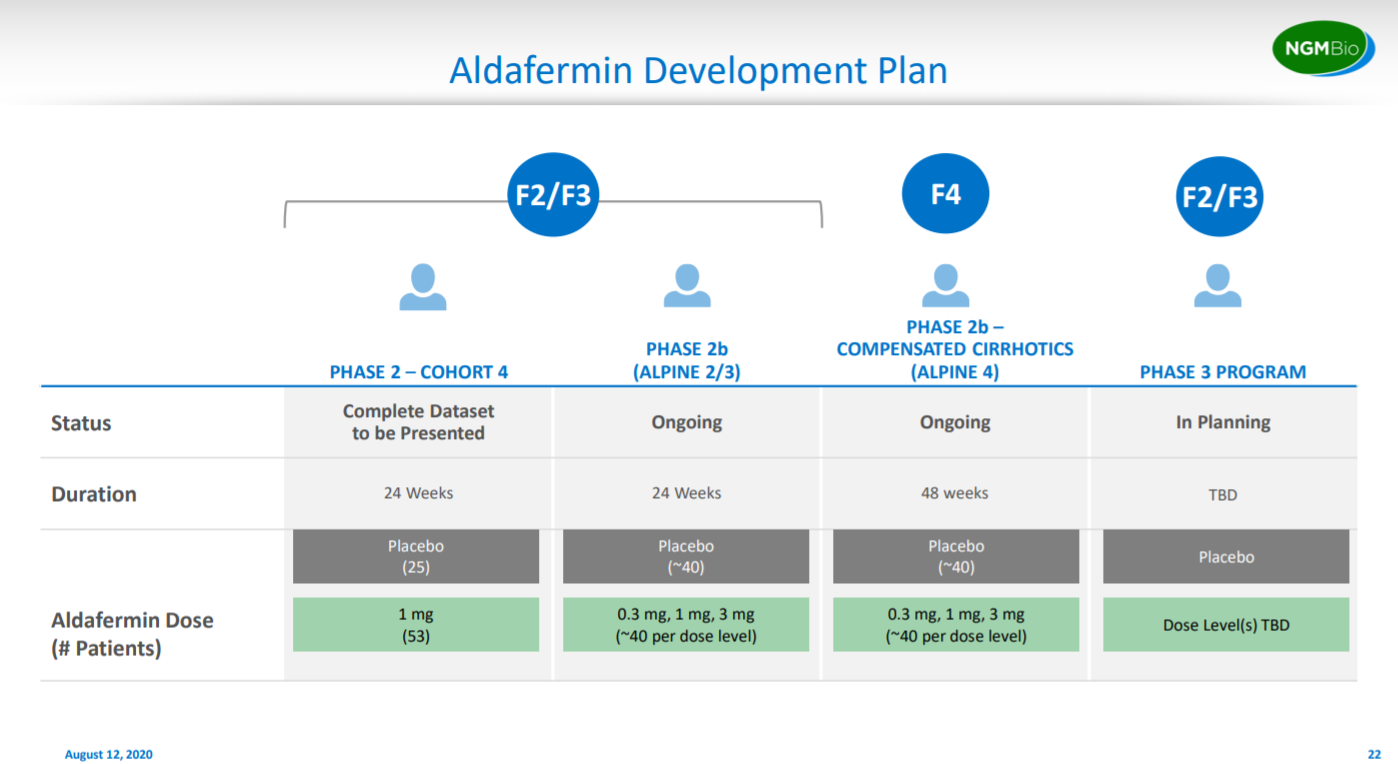
After presenting data on their lead asset, NGM explains their clinical plan in terms of timing and patient numbers to initiate a pivotal study.
The presentation also goes into earlier drug candidates in their pipeline. This slide is once again of NGM’s pipeline.
In particular, NGM maps out their partnership with Merck for earlier-stage assets.
A simple slide to explain the company’s financials. The only metric that kind of matters is runway.
NGM has a catalyst slide that substitutes as one for its milestones.
The deck presents some really exciting data but it could do a better job at explaining its unique business model. The phase 2b data will be exciting to see. If they can get to a pivotal trial for aldafermin, NGM has a shot to transform the lives of NASH patients and beyond.
Follow up questions for the team:
Why do you believe longer treatment duration will improve response rates for Aldafermin?
How does Aldafermin compare to other NASH drugs in development?
Beyond metabolic disease, what other markets is NGM considering? With your internal expertise, the use of structured financing could scale up the business and pipeline.