Axial partners with great founders and inventors. We invest in early-stage life sciences companies such as Appia Bio, Seranova Bio, Delix Therapeutics, Simcha Therapeutics, among others often when they are no more than an idea. We are fanatical about helping the rare inventor who is compelled to build their own enduring business. If you or someone you know has a great idea or company in life sciences, Axial would be excited to get to know you and possibly invest in your vision and company . We are excited to be in business with you - email us at info@axialvc.com
Nevro is a medical device company that has brought their spinal cord stimulation (SCS) to patients suffering from chronic pain. Its Senza system powers Nevro’s HF10 therapy using neuromodulation to clinically show long-term pain relief: https://www.hf10.com/why-hf10/overview/ The product is pretty amazing and Nevro is on the precipice of building a durable business model. Device companies are really hard to get started, but in the long-run, the moats are pretty powerful: there’s really no such thing as a generic medical device.
Founded in 2006, Nevro got backing from Johnson & Johnson, Novo Nordisk, and Covidien and went public in 2014. It took Nevro 9 years from founding to get their product approved in 2015 originally for patients with chronic intractable pain of the trunk or limbs. With the SCS market generating $1B-$2B in revenue per year, Nevro has been able to gain over 15% of the market in ~5 years.
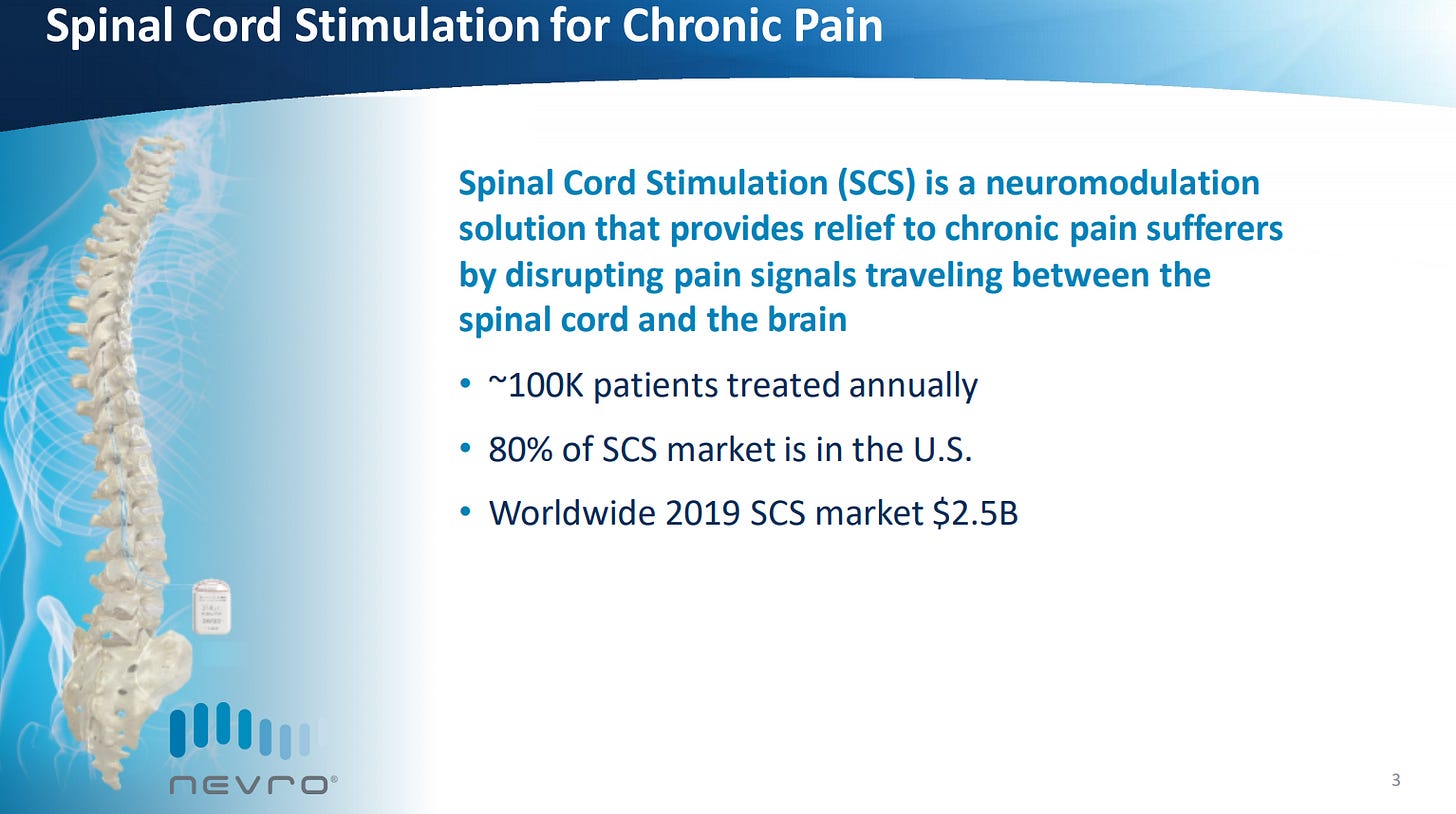
An overview of SCS describing the size of the market in terms of patients.
After an overview, Nevro frames the problem of chronic pain into six parts:
Failed back surgeries are a big reason why people seek out SCS
Around 20%-40% of all back surgeries lead to chronic pain
More than half of all people with chronic pain in the US (50M) have back pain
The fully loaded market cost of chronic pain is $560B (this is not the same as the potential revenue Nevro can earn solving this problem)
Back pain leads to lost productivity
With ~40% of all opioids in the US prescribed for back pain
This is a confusing slide, but Nevro is trying to show that the SCS market is larger than perceived. I am still not sure how the pie chart on the left is different from the one on the right. Maybe adding non-surgical chronic pain might be the market expander.
Company overview
After framing the market opportunity, Nevro shifts into how their HF10 therapy is best suited to solve the problem. The important part is the number of patients implanted with the device - this is often the most powerful moat for any device company.
What makes HF10 unique when compared to other SCS devices is the ability for the device to have an order-of-magnitude higher frequency limit (10 kHz) that can be delivered to a patient’s spinal cord to ensure neural inhibition and pain reduction (measured by responder rates). This is a pretty compelling slide.
To complete their argument, Nevro shows data where 10 kHz can activate inhibitory neurons at a substantially higher levels than 1 or 5 kHz.
Then Nevro shows the clinical work that validated their technology. This is compelling.
From here, the company goes into their history from starting from research at Stanford to confirmatory studies at UC Davis onto clinical studies and approval.
Traction of HF10: after the trial was completed in 2015 and approved, Nevro’s product grew pretty rapidly to over $300M in sales. Impressive.
But sales stalled around 2018 due to salesforce and pricing problems. Nevro does a good job acknowledging these problems.
This slide addresses how Nevro plans to solve problems that arose during commercialization. This first thing that the company did was find a new CEO in March 2019 along with vague salesforce changes.
Commercial drivers
By addressing sales problems, Nevro discusses new products; their Omnia system to deliver a wider range of frequencies to a patient’s spinal cord.
Then they move into the value proposition of Omnia - its ability to deliver more frequencies and hopefully address more of the SCS market.
The product advantage of Omnia is to promote neural inhibition is a wider range of SCS settings.
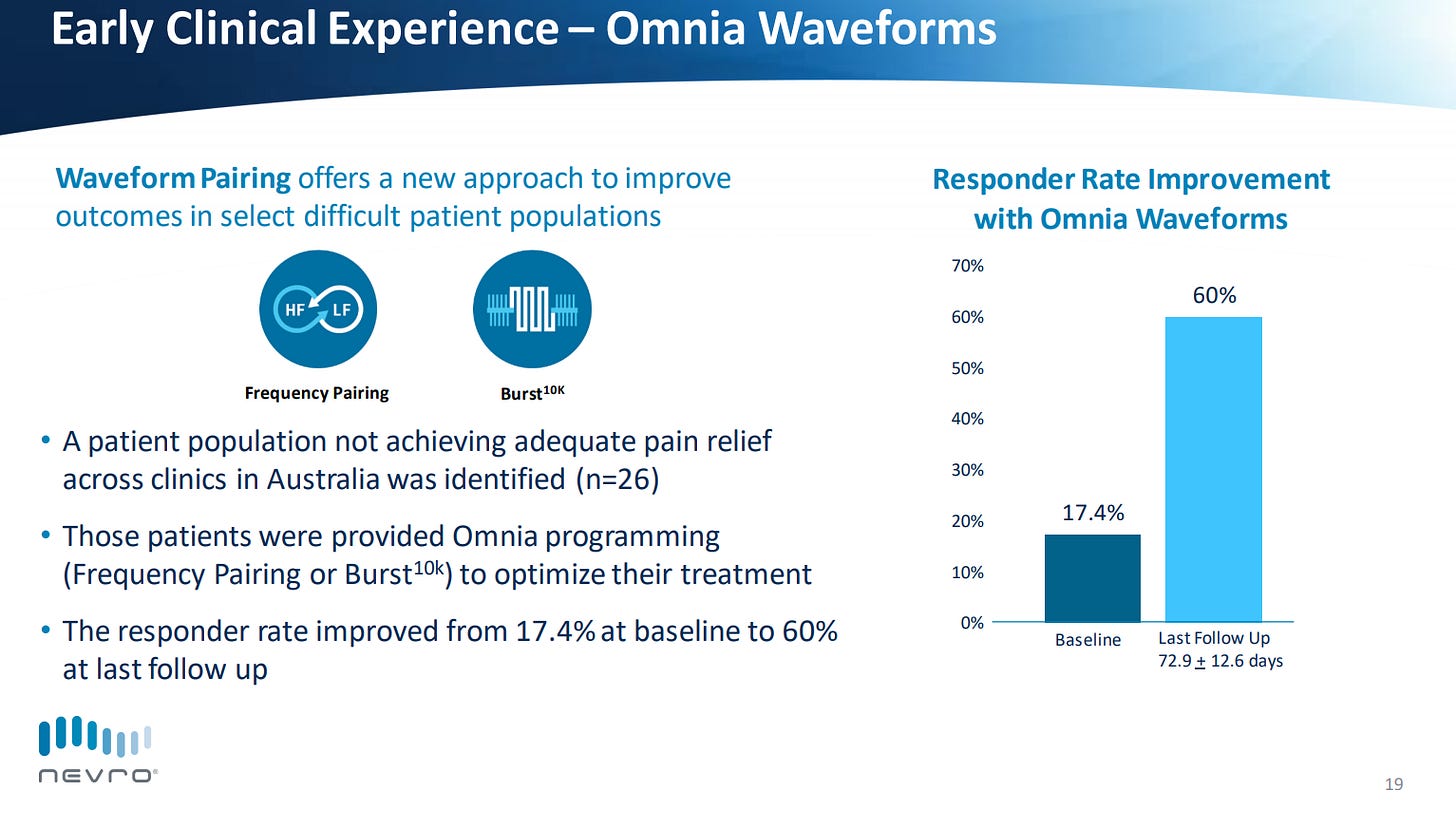
Showing early data to validate Omnia with an ability to transform non-responders to responders. Back to the strategy of expanding their market.
Another product feature Nevro is integrating with Omnia and maybe other systems is a cloud-based platform to track devices and patients.
Then Nevro addresses how these product features build a strong brand around HF10: get patients to request HF10, educate patients on the advantages of HF10, and help them get connected to a provider.
Team and Milestones
The most important task for Nevro is to expand their market.
Painful diabetic neuropathy (PDN) is a major frontier for Nevro with a few million experiencing chronic pain.
This slide addresses the milestones Nevro needs to achieve to help PDN patients: conduct a randomized-controlled study along with follow-up data.
Key clinical events: show data on PDN in 2020 and 2021 along with presenting at various conferences to build up the brand of HF10. The best way to build a strong brand in life sciences is going to conferences and showing data and publishing high-quality papers.
Key commercial events: kickstart sales growth again and introduce their new Omnia product line.
Team: the new CEO is the most important change.
Nevro wraps it all up with the company’s overall strategy:
Expand their addressable market by treat PDN patients and converting non-responders to responders
Realign their salesforce
Rollout a new product
The deck is a little disjointed. It would be easier if they combine their technology slides that feed into their commercial traction and milestones. However, Nevro makes their strategy clear: expand the market and reconfigure their salesforce. Pretty simple.
Like most life sciences businesses, Nevro is in a new phase where pure R&D is no longer the key driver; the company has a salesforce and is in the commercialization phase. The stakes get higher a bit and finding the right balance between product development and sales is tough to get right.
Follow up questions for the team:
In new indications like PDN what is the standard-of-care that could form a barrier to entry for Nevro’s products?
The sales problem is well-characterized, but what specific changes other than getting a new CEO is the company undertaking? Better training? What types of new incentives?
Simply, who are Nevro’s biggest competitors if any?