Axial partners with great founders and inventors. We invest in early-stage life sciences companies such as Appia Bio, Seranova Bio, Delix Therapeutics, Simcha Therapeutics, among others often when they are no more than an idea. We are fanatical about helping the rare inventor who is compelled to build their own enduring business. If you or someone you know has a great idea or company in life sciences, Axial would be excited to get to know you and possibly invest in your vision and company . We are excited to be in business with you - email us at info@axialvc.com
Cabaletta Bio is pioneering the use of chimeric autoantigen receptor (CAAR) T-cell therapies to potentially cure various autoimmune diseases. Autoimmunity is a market that affects 10Ms of people with the standard-of-care often leading to nasty side effects. There is an opportunity to bring technologies that have already been clinically validated in blood cancers by companies such as Kite and Juno to autoimmune diseases.
The company was founded in 2017 out of Penn based on work from the Payne Lab to use clinically-validated CAR-T technologies to target B-cell mediated autoimmune disease. A primary feature of autoimmune disease is the loss of B-cell tolerance and the production of autoantibodies (i.e. they attack self). There are more than 80 autoimmune diseases from rheumatoid arthritis (RA) to multiple sclerosis (MS) to systemic lupus erythematosus (SLE). CAR-T is a technology to engineer T-cells to specifically kill cancer cells. CAR-T has several FDA approvals with multiple billion exits like Kite Pharma and Juno Therapeutics. Cabaletta is essentially repurposing this technology to target autoreactive B-cells instead of cancer cells. Whereas current medicines for autoimmunity kill human B-cells indiscriminately, Cabaletta’s products are engineered T-cells that target solely destructive B-cells.
The first slide of their latest corporate presentation explains their goal to use their CAAR-T technology to be the first to cure an autoimmune disease.
After the mission slide, it’s always useful to give some background on the business or product development or both. Cabaletta uses a bullet point to go over their technology then elaborates on their pipeline status and runway. Early-stage startups probably should leave the latter out.
Then Cabaletta goes into how CAAR-T compares to CAR-T. The former presents an autoantigen to target a B-cell whereas the latter presents an antibody fragment to target a cancer cell. This is the core premise of what makes Cabaletta unique; they essentially just swap out a part of the effector construct to target a different cell type than CAR-T. Given that CAR-T has seen clinical success, Cabaletta’s CAAR-T technology has good odds of seeing similar success.
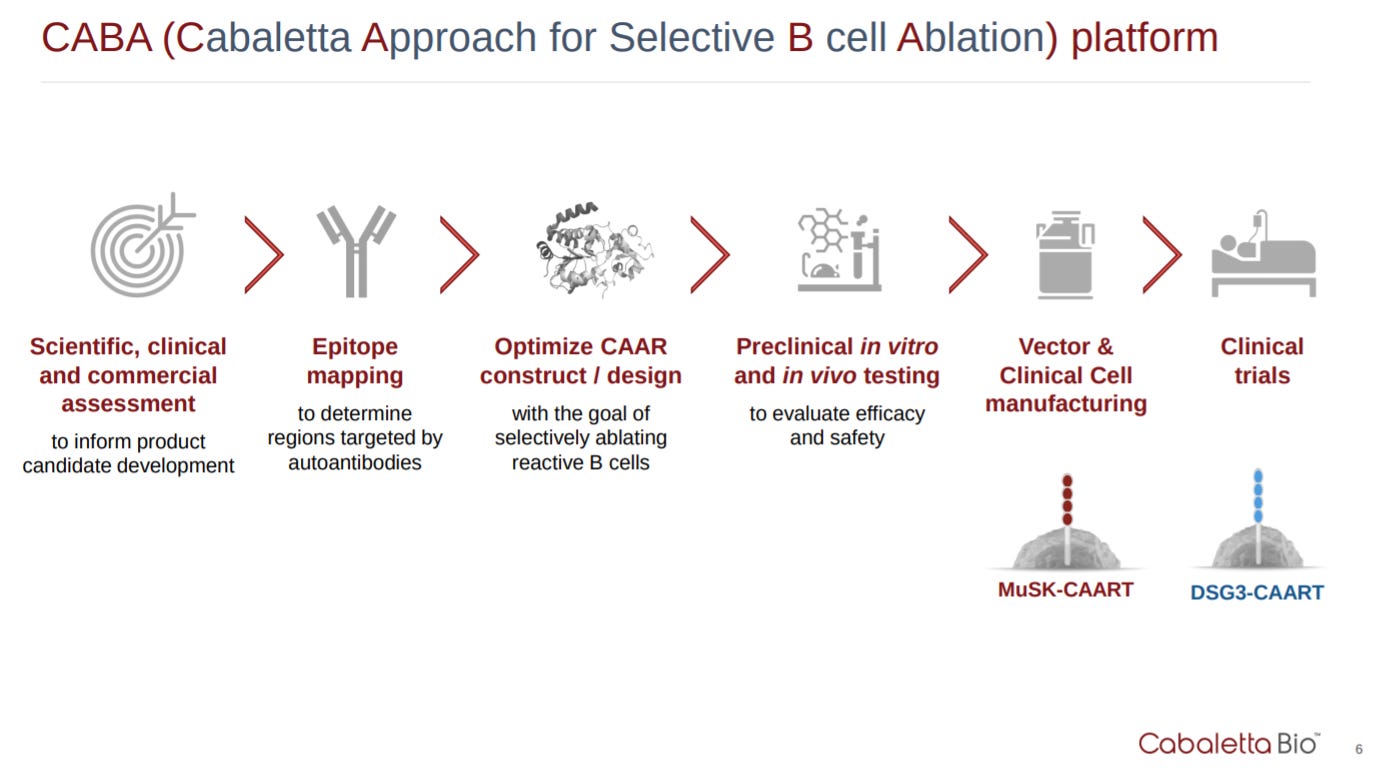
Then the company lays out their platform. The key insight is that cell and gene therapy companies need to be vertically integrated (i.e. Arsenal Bio, Lyell).
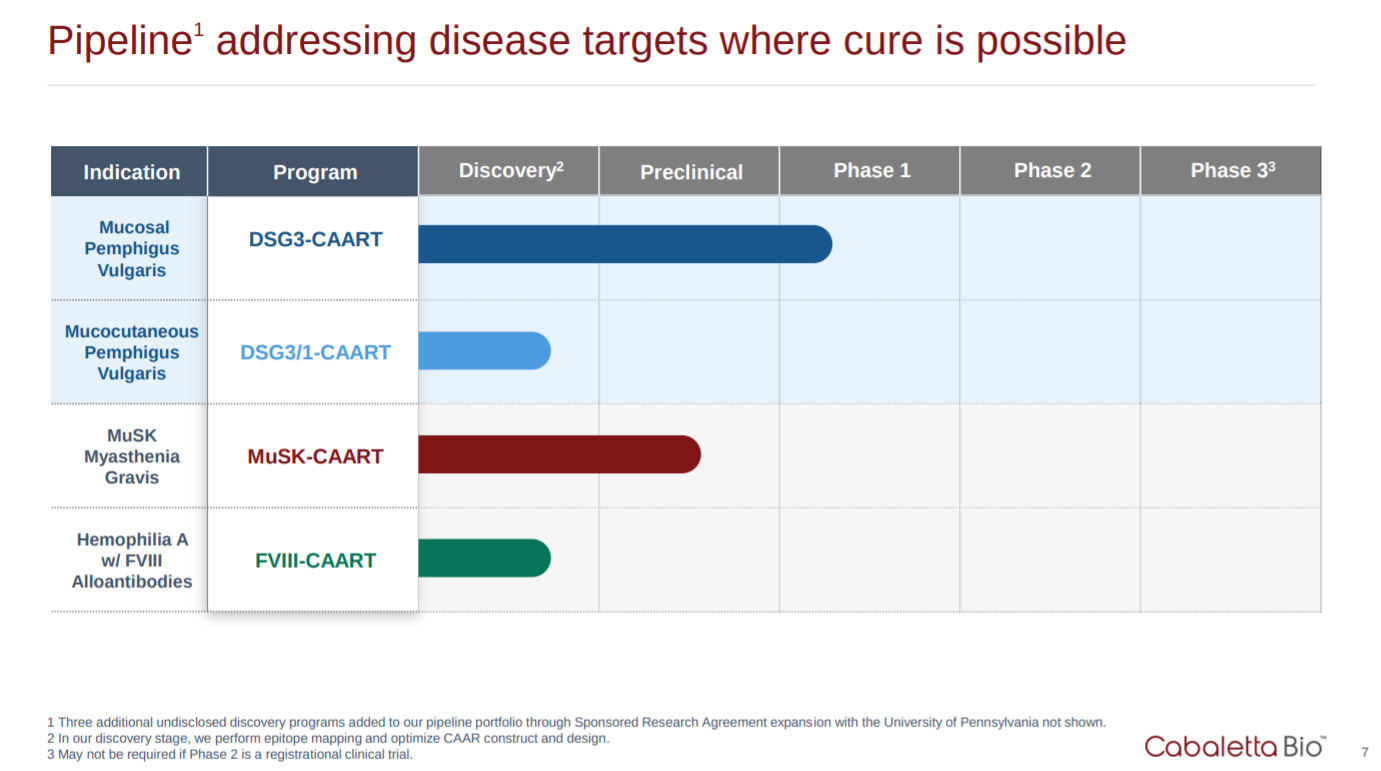
After touching upon their technology, Cabaletta has the standard pipeline graphic. CAAR-T is a new modality and they could do a better job at conveying how they can own the entire space especially from an IP perspective. Look at Alnylam for RNAi and maybe Regeneron for antibody humanization in mice.
After laying out their technology and potential products, the company goes into their lead asset.
This is a really important slide where Cabaletta explains their logic to choose the first indication: mucosal PV. For every science-driven company, choosing the right first market is always hard. The market sizes are often false signals; the underlying logic to choose that market should also be driven by the biology and any clinical validation.
Then Cabaletta gives an overview of pemphigus vulgaris. This is a pretty good template to overlay the standard of care with the market size.
Cabaletta moves into the design of their construct. They focus on presenting a series of autoantigens that comprehensively cover autoantibodies specific for DSG3 on autoreactive B-cells.
With this, Cabaletta explains their preclinical data in terms of safety and target engagement. Pretty exciting results.
With preclinical data on their lead asset, Cabaletta explains the design of the phase 1 trial they have initiated.
Phase 1 trials are focused on safety. So Cabaletta lays out the risks of the trial along with solutions to 3 major problems they may encounter: neurotox, dosing problems, and flare ups. This slide also alludes to endpoints: measuring safety at day 8 and 90 along with measuring target engagement after 6 months.
This slide focuses on a key risk around dosing. Patients enrolled might have inhibitory antibodies are concentrations that reduce CAAR-T target engagement and weirdly expand CAAR-T proliferation. It might be worth it to do an immunoassay to recruit particular patient populations, but then that might reduce the market opportunity.
The company shows that in a mouse model with inhibitory antibodies at physiological levels for humans, their CAAR-T candidate shows target engagement. All of this preclinical data is what you call IND-enabling. Then you got to do the experiment in humans.
This slide touches on an important point that lymphodepletion might not be useful in an autoimmune setting. They could do a better job at explaining why and the possible advantages in terms of the potential higher dosing in autoimmune patients. The company also has a few slides on their other assets in preclinical development focused on MuSK and mcPV.
With the technology and pipeline explained, every cell therapy company is only as good as its manufacturing plan. The process is the product. Cabaletta is initially going to rely on Penn and CHOP to manufacture its CAAR-T cells for the phase 1 then start using CDMOs for the scale up with the end-goal to control manufacturing themselves.
With this plan, the company compares the manufacturing for CAAR-T versus CAR-T. In short, CAR-T manufacturing targeting CD19 and its ramp up derisks Cabelleta’s process; the company knows what protocols will get FDA approval.
The penultimate slide is on the team. The inventor, Aimee Paye has an important role as co-chair of the SAB.
Like every good biotech company, the last slide is around milestones. For their lead product, Cabaletta wants to get a first read out on their phase 1 trial by 1H21 and get their second product to IND by 2H21.
The deck does a great job to show how success in CAR-T for oncology is helping Cabaletta have a potentially higher probability of success in autoimmunity. The phase 1 data release in early 2021 could bring validation to the strategy and open up the field of targeted cell therapies in autoimmunity.
Follow up questions for the team:
What criteria are patients recruited for the phase 1 trial?
What is the plan for the platform to expand to more indications? Adding technologies to construct design? Investing in more R&D to map out targets for autoreactive B-cells.