Axial partners with great founders and inventors. We invest in early-stage life sciences companies such as Appia Bio, Seranova Bio, Delix Therapeutics, Simcha Therapeutics, among others often when they are no more than an idea. We are fanatical about helping the rare inventor who is compelled to build their own enduring business. If you or someone you know has a great idea or company in life sciences, Axial would be excited to get to know you and possibly invest in your vision and company . We are excited to be in business with you - email us at info@axialvc.com
STING
A research group at Merck led by George Addona recently published a paper that developed an orally available STING agonist. The lead author, Bo-Sheng Pan, had a distinguished career at Merck over ~3 decades in pharmacology and cancer research and retired around 2 years ago. The paper used a phenotypic screen to identify a small molecule, MSA-2, that non-covalently activates STING (stimulator of interferon genes). STING is an endoplasmic reticulum (ER) transmembrane receptor protein that is part of a signaling pathway that results in upregulation of molecules that are part of the innate immune response. Upon sensing dsDNA, the enzyme cyclic-GMP-AMP-synthase (cGAS), cyclizes GTP and ATP to produce the second messenger cyclic-GMP-AMP (cGAMP), which binds to and activates STING.
This paper is a major breakthrough in the field of innate immunity - the first-generation of STING inhibitors have been directly injected into the site of the tumor to avoid degradation. The key insight for why MSA-2 exhibits this property is that it exists in equilibrium between monomers and dimers (the dimer form activates STING) favoring the monomer state. This enables specificity to tumor sites and higher stability because MSA-2 is more likely to dimerize in the acidic conditions of the tumor microenvironment.
A pivotal moment in the STING field came in 2013 when the Chen Lab at the University of Texas Southwestern Medical Center discovered that an enzyme called cyclic GMP-AMP synthase (cGAS) binds DNA then produces cGAMP to activate STING. This is another monumental paper in the innate immunity field: this was a keystone moment to generate billions of dollars in investments to develop cGAMP analogs to activate STING to treat cancer. However, cyclic dinucleotides are quickly broken down in the human body by phosphodiesterase enzymes. So the first generation of STING agonists required direct injection to the tumor site, a major limitation to convert these candidates into new medicines. As a result, the next generation of STING agonists will need to be orally available or at least be given through an IV.
Key experiments:
Phenotypic screening of over 2M compounds to find chemical matter with the potential to activate STING and have desirable permeability features.
The screening process discovered a hit MSA-2. Mouse models were used to test MSA-2 anti-cancer effects, and the rechallenge experiment conducted verified the molecule’s durable effects.
In search of MSA-2’s mechanism to agonize STING, a covalent dimer of the compound was synthesized to confirm that dimerized MSA-2 selectively activates STING.
What milestones are required to design a business or commercial project around this invention:
More optimization and medicinal chemistry work to improve PK/PD properties of MSA-2. Prodrugs and conjugation can also be explored here to selectively activate the drug candidate in specific tissues.
Exploring whether a STING agonist can be successfully used as a monotherapy for specific cancer indications?
Verifying that the oral delivery route of MSA-2 does not induce systemic STING activation, which will lead to toxicity in humans.
Generating and developing analogs of MSA-2.
The group developed a phenotypic screen measuring interferon-b (IFN-b) secretion in THP-1 (an immortalized monocyte cell line) cells as a way to detect STING agonism. With this method, around 2.4M small molecules were screened: MSA-2 along with a few other hits were found to induce IFN-b secretions. To confirm MSA-2’s agonism of STING:
The small molecule also induced IFN-b production in mouse macrophages
MSA-2 was found to disrupt binding of cGAMP to STING biochemically
In THP-1 cells knocked out for STING, MSA-2 failed to induce IFN-b production
TBK1 and IRF3, proteins downstream from STING, were both phosphorylated in cells treated with MSA-2
With a promising hit, the group tested the pharmacokinetics and pharmacodynamics (PK/PD) of MSA-2 in vivo through 3 delivery routes in a colon carcinoma mouse model:
Intratumoral (IT)
Subcutaneous (SC)
Oral (PO)
In Figure 3B/C (shown below), MSA-2 delivered through SC and PO had similar pharmacokinetics (i.e. bioavailability) in both the tumor and plasma. In vivo, MSA-2 showed a dose-dependent anti-tumor effect (Figure 3D/E/F). This effect persisted in rechallenged mice (Figure 3K). In cancer models, rechallenging is probably the most important experiment to verify that the anti-tumor effect of MSA-2 is durable enough to initiate a drug development process. In mice without detectable STING, MSA-2’s anti-tumor activity was not observed. These set of experiments showed that orally-available MSA-2 had substantial effects on a cancer model in a STING-dependent manner.
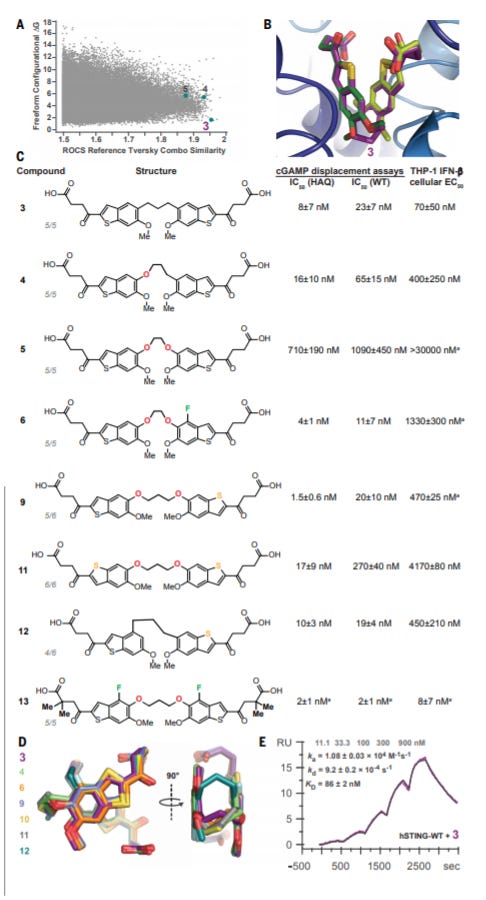
The next step was to determine the mechanism by which MSA-2 activates STING selectively. X-ray crystallography was used to determine the structure of MSA-2 bound to human STING (In A for the image below), and nuclear magnetic resonance (NMR) was used to measure the open and closed (binding to MSA-2) conformations of STING. With this starting point the group created 3 hypotheses (part C) to test:
MSA-2 as a monomer binds two sites of the STING dimer independently
MSA-2 (monomeric) binding one site on STING alters the affinity for MSA-2 to the other site
In equilibrium between monomers and dimers, MSA-2 can only bind STING as a dimer
For model 1, the Hill coefficient (measuring the binding of ligands to a target as a function of ligand concentration) was not compatible (part D) with independent ligand binding. Surface plasmon resonance was used to determine the kinetics of MSA-2 binding to STING discovering that the data was incompatible with model 2. NMR was used to compare MSA-2 binding to a control compound to figure out that the former can be found dimerized.
With the model that MSA-2 dimerized is the agonist for STING, a covalent dimer of MSA-2 was synthesized and shown to generate similar effects as MSA-2 in its original state. The group used a computational method to score thousands of linkers to score the new MSA-2 dimer for the reduction in entropy and maximize the alignment of the crystal structure of MSA-2 bound to STING (part A of the image below). Dimer 3 (part C) was synthesized, along with other candidates, and was found to agonize STING (part E). This experiment combined with the observation that MSA-2 exists as a monomer and dimer confirms the third model suggested by the group.
The final experiment was to see if MSA-2 anti-tumor effects are synergistic with a checkpoint inhibitor. The group combined MSA-2 with an anti-PD-1 antibody in 4 tumor models:
B16F10 as a melanoma model (part A of the image below)
LL-2 as a lung cancer model (part B)
Advanced MC38 for colorectal cancer (C)
CT26 for colorectal as well (D)
In each of the models, MSA-2 given orally or subcutaneously and the anti-PD-1 antibody delivered intraperitoneally synergized to increase overall survival. Other readouts were:
In the MC38 model, tolerability of MSA-2 in terms of body weight was dose dependent
In the LL-2 model, combining MSA-2 with the checkpoint inhibitor increased CD8+ T-cell infiltration in the cancer
In the MC38 model, models that are either T-cell deficient or immunodeficient did not respond to MSA-2 as robustly just confirming the molecules mechanism-of-action (MoA) acting through the innate immune system
With two key experiments - the rechallenge test and showing that dimerized MSA-2 agonizes STING, the paper developed MSA-2 as an orally-available drug candidate to activate STING to drive tumor regression.
Ultimately, the Merck group used a high-throughput cell-based phenotypic screen to discover a potential STING agonist. MIce studies confirmed the hits anti-tumor effects when orally delivered and biochemical studies honed in on its MoA. STING is a master regulator of type I interferons and an important player in the human immune system - to spart the innate immune system and enhance the adaptive arm. In short STING acts as a bridge between the two parts of the immune system. This role makes STING a promising target to pursue currently drug resistant tumors.
Past drug discovery programs focused on developing analogs of cyclic dinucleotide (CDN) agonists. However, these molecules have poor bioavailability and lead to off-target effects when delivered systemically, and therefore need to be delivered intratumorally. The identification of MSA-2 as an orally available, non-CDN STING agonist is a promising discovery in cancer drug development. A lot more work in terms of optimization and the basic biology of STING is still required to fully realize the potential of the pathway for patients.