Axial partners with great founders and inventors. We invest in early-stage life sciences companies such as Appia Bio, Seranova Bio, Delix Therapeutics, Simcha Therapeutics, among others often when they are no more than an idea. We are fanatical about helping the rare inventor who is compelled to build their own enduring business. If you or someone you know has a great idea or company in life sciences, Axial would be excited to get to know you and possibly invest in your vision and company . We are excited to be in business with you - email us at info@axialvc.com
Sana Biotechnology is a cell therapy company, recently filing an S-1, founded in 2018 to build a platform for in vivo and ex vivo cell engineering. The company was built to bring these technologies to cancer but diabetes, central nervous system (CNS) disorders, cardiovascular diseases, and genetic disorders as well. The company’s pipeline is still preclinical with the expectation of an investigational new drug application (IND) in 2022/2023.
Important problems to solve in order to realize the full potential of cell therapies are:
Manufacturing
Engraftment
Function
Persistence and overcoming rejection from the human immune system
As a result, most cell therapy companies have focused on autologous cell therapies (i.e. from the patient’s own cells) to avoid host rejection. However, autologous therapies have a complex manufacturing process that has been difficult to scale and reduce the cell types that can be currently used. To avoid this problem, Sana is centered around technology to generate hypoimmune (i.e. avoid detection from the human immune system) cell therapies from iPSCs or an allogeneic cell particularly for T-cells.
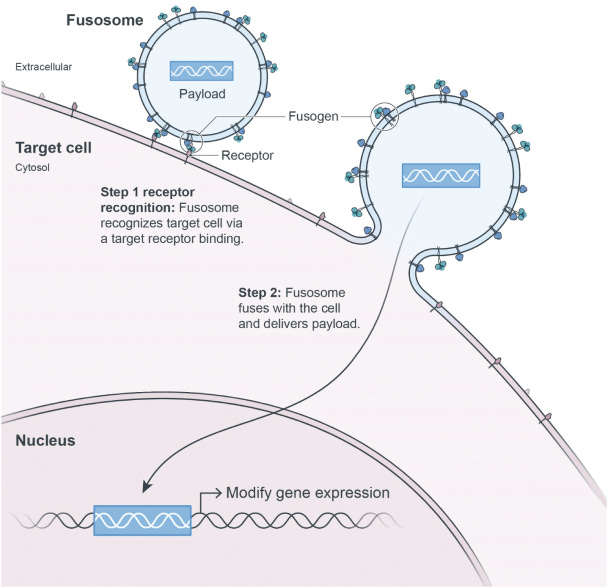
Sana recognizes that delivery to these hypoimmune cells are their greatest challenge. They use their fusogen technology to selectively deliver vehicles through specific cell surface receptors (like CD8) that create fusosomes and deliver a payload into a cell. Sana’s initial focus is on T-cells, hepatocytes, and hematopoietic stem cells (HSCs); however, their technology could expand to glia cells and beta islets. By combining a diverse toolkit from gene editing, protein engineering, stem cells, and immunology, the company is bringing breadth to the problem of cell therapy engineering and manufacturing.
Highlights
ARCH and Flagship dominate the cap table since they put up a few $100M to get Sana started. Steven Harr (CEO) still owns a pretty significant amount (over 5%) given that the company raised so much capital ($700M) from day one.
Sana’s two most advanced drug candidates are hypoimmune CAR-T cell therapies, SC291 and SC255 - SC291 targets CD19 for NHL, CLL, and ALL and SC255 targets BCMA for multiple myeloma. These candidates face 2 issues: (1) graft versus host disease (GvHD) and (2) host versus graft disease (HvGD). For the former, cells can be edited to delete TCR components. For the latter, Sana brings their hypoimmune technology versus the standard to use drugs to eliminate a patient’s immune system.
Sana’s 3 main markets are oncology, diabetes, and CNS disorders. NHL, CLL, and ALL affect around 70K patients per year. T1DM affects around 1.6M people in the US and 2.4M in Europe. For the CNS disorders Sana is addressing, multiple sclerosis affects approximately 1.6M people and Huntington’s disease affects approximately 90,000 people in the US.
Team
Sana’s Co-Founder, President, and CEO is Steven Harr. He was the former CFO and Head of Corporate Development at Juno Therapeutics and previously was a managing director and Head of Biotechnology Investment Banking at Morgan Stanley. Sunil Agarwal is also a Co-Founder of Sana and is the Chief Medical Officer. Agarwal was the President of R&D at Juno, a former partner at Sofinnova Ventures, and an EVP at Ultragenyx Pharmaceutical.
Richard Mulligan heads SanaX, the R&D division of Sana. He is a professor emeritus at Harvard Medical School. While he was a professor, Mulligan advised Icahn Capital in biotechnology investments and was a founding partner at Sarissa Capital Management. Christian Hordo is the Chief Business of Sana where he previously was a VP Juno and a team leader at Genentech.
Investors
ARCH and Flagship dominate the cap table since they put up a few $100M to get Sana started. Steven Harr (CEO) still owns a pretty significant amount (over 5%) given that the company raised so much capital ($700M) from day one.
Technology
The core technology for Sana is around the company’s ability to generate hypoimmune cells:
“Disruption of MHC class I and class II expression (which inactivates adaptive immune responses), and overexpression of CD47 (which hides cells from the innate immune system, including macrophages and natural killer (NK) cells)”
“Pluripotent stem cells from healthy donors are used as the starting material and are then genetically modified with the hypoimmune edits”
“These edited cells are then differentiated into cell types of therapeutic interest, which are administered to the patient as ‘off the shelf’ therapies”
Sana has demonstrated the ability of a diverse set of hypoimmune cells to avoid immune rejection in model organisms. Combining this ex vivo tool with in vivo delivery has the potential to modify a broader range of cells and treat diverse sets of patients. Sana’s initial focus is on T-cells (CD8+ and CD4+) targeting CD19 in oncology from NHL to CLL and ALL. The longer term goal is to expand to more targets like BCMA and go beyond cancer into chronic disease and CNS disorders.
Sana’s two most advanced drug candidates are hypoimmune CAR-T cell therapies, SC291 and SC255 - SC291 targets CD19 for NHL, CLL, and ALL and SC255 targets BCMA for multiple myeloma. These candidates face 2 issues: (1) graft versus host disease (GvHD) and (2) host versus graft disease (HvGD). For the former, cells can be edited to delete TCR components. For the latter, Sana brings their hypoimmune technology versus the standard to use drugs to eliminate a patient’s immune system.
Sana is also developing pancreatic beta cells, SC451, for the treatment of diabetes focusing on type I diabetes mellitus (T1DM). The company is also developing stem cell-derived allogeneic glial progenitor cells (GPC) that can differentiate into myelin-producing oligodendrocytes or astrocytic support cells for neurons to treat MS, Huntington’s disease, and myelin diseases.
Market
Sana’s 3 main markets are oncology, diabetes, and CNS disorders. NHL, CLL, and ALL affect around 70K patients per year. T1DM affects around 1.6M people in the US and 2.4M in Europe. For the CNS disorders Sana is addressing, multiple sclerosis affects approximately 1.6M people and Huntington’s disease affects approximately 90,000 people in the US.
Overall, cell transplantations have been a powerful tool in medicine. However, immune rejection has been a barrier to bring this tool to more diseases:
“Studies established T cells as playing a key role in the host immune response to transplant. T cells belong to the adaptive immune system, recognizing and eliminating non-self cells via recognition of differences in cell-surface proteins encoded by the major histocompatibility (MHC) locus. There are two types of MHC molecules: MHC class I, expressed on the surface of almost all nucleated cells, and MHC class II, expressed constitutively on professional antigen presenting cells (APC), including macrophages and dendritic cells . Expression of MHC class II is also induced in many additional cells in the context of inflammation. MHC class I molecules typically display peptides on the cell surface from degraded intracellular proteins. Cells display peptides from normal self proteins on MHC class I, which typically will not activate an immune response due to a process called tolerance, where the body recognizes these peptides as self . However, if a cell displays a peptide from a foreign or mutated protein on MHC class I, for example as a result of a protein mutation, it may result in the activation of a cytotoxic T cell response specific to the peptide-MHC complex via the T cell receptor (TCR) on the T cell surface. The activated T cell then eliminates the cell
MHC class II molecules typically display peptides derived from phagocytosis of extracellular proteins on the surface of APCs. These peptide-MHC complexes interact with TCRs on helper T cells, such as CD4+ T cells, resulting in a downstream cellular and humoral immune response. The humoral immune response leads to antibody production against foreign proteins
In allogeneic transplants, the cellular and humoral processes can recognize proteins from the donor as ‘foreign’, resulting in an immune response to the transplant including potential elimination of the transplanted cells. In the allogeneic setting, MHC proteins can be highly immunogenic due to their inherent polymorphism, increasing the risk of the recognition of transplants as foreign. This underlies the basis for MHC typing and matching to assess and reduce the risk of organ transplant rejection.”
Other companies have focused on downregulation or deleting MHC. This approach does not help a cell evade the innate immune system. “Natural killer (NK) cells express receptors known as inhibitory killer-cell immunoglobulin-like receptors (inhibitory KIRs). KIRs recognize self MHC class I molecules on the surface of cells and provide inhibitory signals to the NK cells to prevent their activation. Cells missing MHC class I molecules are corresponding eliminated by NK cells because of the lack of inhibitory KIR signaling and a resulting cytolytic activation.”
Sana’s hypoimmune cells are working to avoid both the adaptive and innate immune response. This work was derived from studying fetomaternal tolerance during pregnancy: “The fetus, despite having half its genetic material from the father, is not rejected by the mother’s immune system. However, after birth, few if any children would qualify as a matched donor for a cell or organ transplant for their mother These scientists categorized the differences of the maternal-fetal border and systematically tested them to understand which, if any, of these were most important to immune evasionThey have tested these changes in both in vitro and in vivo animal models.”
Business model
Sana is building a fully-integrated cell therapy company. Their approach has been to develop technologies in house and also acquire them, as with the fusogen IP, in a land grab strategy:
“Minimize biology risk where there is platform risk, or in other words, prioritize opportunities where success with our platform should lead to success in addressing the underlying disease.” This is expressed by Sana’s focus on CD19, a derisked target in oncology, to get to clinical validation with the goal to expand to newer targets.
“Prioritize program investments in diseases where the strengths of our in vivo and ex vivo platforms can address key limitations of existing therapeutic approaches”
“Focus on conditions of high unmet need, including the most grievous diseases”
“Prioritize efforts where success in one area begets success in others”
Over time, Sana wants to expand to more cell types and diseases while brining different modalities to engineer those cells.
Valuation
Sana’s current valuation is mainly in its platform. Without any clinical assets, the company’s value is really in its future ability to develop cell therapies that can be manufactured more easily and for more diseases than incumbents:
“Delivery - we believe the critical limitation for in vivo cell engineering is delivery, and therefore, we are investing significantly in delivery technologies, including our fusogen technology, which is designed to enable both cell-specific delivery and delivery of diverse payloads”
“Gene modification - there has been substantial recent progress in gene modification and the field is now at the point where virtually any desired modification can be performed in vitro. However, no single technology or platform is optimal for all possible applications.To this end, we are developing capabilities across multiple technologies and investing to develop our own novel technologies to be applied on a case-by-case basis.”
“Manufacturing - we are investing proactively in process development, analytical development, CMC regulatory, and other manufacturing sciences in order to enable scalable manufacturing of our in vivo therapies and ensure broad access”
Mention highlights in the S-1
Hypoimmune: 3188
iPSC: 101
Allogeneic: 95
Flagship: 78
Safety: 78
Scale: 35
SanaX: 32
Alliances: 8