Axial partners with great founders and inventors. We invest in early-stage life sciences companies such as Appia Bio, Seranova Bio, Delix Therapeutics, Simcha Therapeutics, among others often when they are no more than an idea. We are fanatical about helping the rare inventor who is compelled to build their own enduring business. If you or someone you know has a great idea or company in life sciences, Axial would be excited to get to know you and possibly invest in your vision and company . We are excited to be in business with you - email us at info@axialvc.com
NexImmune is a biotechnology company developing new immunotherapies in acute myeloid leukemia (AML) and multiple myeloma (MM) that recently filed a S-1. The company has two lead drug candidates in clinical trials:
NEXI-001 in AML
NEXI-002 in MM
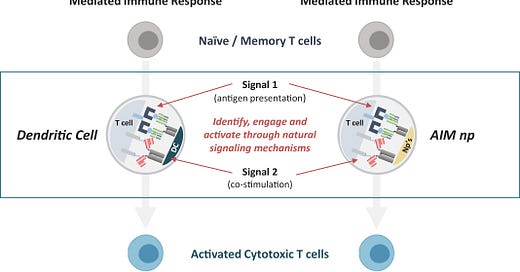
The company is centered around their AIM (Artificial Immune Modulation) platform that relies on nanoparticles to activate T-cells similar to a synthetic dendritic cell. The technical premise is to direct T-cell killing without having to do any cellular engineering.
Highlights
“[The] AIM nanoparticle technology is designed to bypass the dendritic cells and deliver the right kind of instructions directly to T cells using natural biology. In essence, [Neximmune] creates nano-sized synthetic dendritic cells. These nano-sized synthetic dendritic cells are designed to deliver precise instructions to a specific set of targeted T cells, and these instructions will be different depending on the therapeutic goal.”
Ultimately, the modular design of AIM could give Neximmune the potential to expand to other cancer and immunological diseases. With the ability to use nanoparticles in place of antigens, the company plans to focus on blood cancers then move to solid tumors onto autoimmunity and infectious disease.
The company’s business is a platform creating a pipeline of drug candidates. Neximmune is positioning themselves to use AIM to generate templates for immunotherapy combination treatments and off-the-shelf T-cell products in cancer and other diseases.
Team
Neximmune’s CEO is Scott Carmer. He was previously the company’s COO and CBO and worked at MedImmune leading commercial operations. Carmer was VP of Sales & Marketing at Genentech and used to work at Amgen and GSK. Jerome Zeldis is Neximmune’s EVP of R&D who was previously the CMO at Sorrento Therapeutics and Celgene. The company’s COO is Kristi Jones who was an VP at AstraZeneca/MedImmune, and Neximmune’s CMO is Robert Knight was a VP at Kite Pharmaceuticals and used to work at Sorrento and Celgene as well.
Investors
The cap table is dominated by ex-Celgene people, non-bluechip funds, and Allen & Company.
Technology
“At the center of the immune response are T cells, often referred to as the ‘foot soldiers’ of the immune system. Whenever healthy cells are under attack, either by a virus, bacteria or cancer, the immune system calls on the T cell to identify, engage and kill the specific invader or diseased cells. Importantly, natural T cells have the ability to distinguish between diseased and healthy cells. However, T cells need very specific sets of instructions to function effectively. In healthy individuals, these specific instructions are normally delivered to the T cells by dendritic cells, which are also referred to as professional antigen-presenting cells. Dendritic cells provide these instructions through key signaling proteins. However, cancer cells often compromise the function of dendritic cells and the instructions they deliver to T cells.
[The] AIM nanoparticle technology is designed to bypass the dendritic cells and deliver the right kind of instructions directly to T cells using natural biology. In essence, [Neximmune] creates nano-sized synthetic dendritic cells. These nano-sized synthetic dendritic cells are designed to deliver precise instructions to a specific set of targeted T cells, and these instructions will be different depending on the therapeutic goal. Translating this to cancer, each infusion, or product, contains populations of T cells that can identify and attack multiple tumor-specific antigen targets on a tumor cell. In preclinical studies, [Neximmune has] observed that AIM-activated T cells were potent, were able to effectively distinguish between tumor cells and healthy cells, and should have potential for long term persistence.”
The AIM platform relies on 3 key principles:
“Precision - delivering specific sets of instructions to specific T cell populations that direct a specific T cell function”
“Potency - Direct T cells to attack multiple disease relevant antigen targets through naturally occurring identification, engagement and killing mechanisms, with reduced potential for undesired toxicities”
“Persistence - Maintain T cell subtypes that support self-renewal, proliferation, immunologic memory and long-term T cell survival”
“The AIM technology is similar to other therapeutic approaches that are using naturally occurring, but unselected, tumor-infiltrating lymphocytes, or TILs. Current TIL therapies have generated impressive clinical responses in difficult-to-treat patient populations, including patients with relapsed/refractory solid tumors like melanoma and cervical cancer. However, TIL products contain a significant range of variability from one product to another, including preferred ratios of CD8+ and CD4+ T cells, optimal T cell phenotypes, and known T cell tumor-specificity. Given currently-employed TIL isolation and product manufacturing processes, it is not possible for current TIL therapies to control for any one of these important product attributes. Because our AIM technology uses synthetic dendritic cells designed to deliver specific instructions directly to specific populations of T cells, combined with a well-controlled manufacturing process, we believe we can produce product candidates with highly consistent in vitro characteristics that are associated with clinical responses: CD8+ T cells, T stem cell-like and memory subtypes, and antigen-specific recognition.
Genetically engineered T cell approaches, such as chimeric antigen receptor T cells, or CAR Ts, or engineered T cell receptor, or TCR, technologies are very precise in their engineering, but have the key limitation of single antigen targeting, and have been associated with life-threatening side effects and limited durability. The manufacturing processes currently used for the in vitro activation and expansion of engineered T cell products results in T cell products that contain high proportions of terminally differentiated and exhausted T cell subtypes, with limited potential for in vivo persistence.
A theoretical advantage of engineered T cell products is enhanced antitumor potency, achieved through the transduction of high affinity and/or affinity-enhanced TCRs or through re-engineering the entire TCR complex. [Neximmune has] conducted in vitro experiments to compare the potency of both TCR-engineered and CAR T-transduced products with [their] AIM-activated T cell products. In tumor cell-line killing assays designed to assess the activity of our AIM-activated T cells with that of TCR and CAR T products, [their] AIM-activated T cells showed killing potency comparable to both genetically engineered modalities.”
Neximmune has 2 clinical stage drug candidates: NEXI-001 and NEXI-002. Both are adoptive T-cell therapies (ACTs) with CD8+ T-cells. “NEXI-001 is a donor-derived, or allogeneic, ACT in a Phase I/II clinical trial for the treatment of patients with relapsed AML after allogeneic stem cell transplantation, or allo-HSCT. NEXI-002 is a patient-derived, or autologous, ACT in a Phase I/II clinical trial for the treatment of MM patients that have failed at least three prior lines of therapy. In December 2020, initial safety, tolerability and immunologic data from our NEXI-001 trial was shared as an oral presentation during the 62nd American Society of Hematology (ASH) Annual Meeting. These preliminary data showed that single infusions of NEXI-001 T cells in the first three patients treated were well-tolerated. and [Neximmune] observed initial indicators of immunologic response after NEXI-001 T cell infusion in each of the three patients dosed, including (i) lymphocyte reconstitution to pre-lymphodepletion baseline levels at timepoints early within the expected range, and an earlier-than-expected recovery of the CD4+ T cell compartments; (ii) the presence, proliferation and persistence of NEXI-001 antigen-specific T cells as measured in peripheral blood; (iii) clonal expansion of NEXI-001 T cells in both peripheral blood and bone marrow; and (iv) the persistence of T cell subtypes present in NEXI-001 product candidates over time, as measured in peripheral blood. It is important to note that [Neximmune is] early in the safety evaluation and dose-finding part of the Phase I/II trial, and that these results are derived from the first three patients only and are not statistically significant. [Neximmune expects] to announce initial data for most patients in both the NEXI-001 and NEXI-002 clinical trials by the end of 2021.
Ultimately, the modular design of AIM could give Neximmune the potential to expand to other cancer and immunological diseases. With the ability to use nanoparticles in place of antigens, the company plans to focus on blood cancers then move to solid tumors onto autoimmunity and infectious disease.
Market
Neximmune’s strategy is to establish a proof-of-concept in blood cancers to validate the AIM platform. After, the company wants to expand to solid tumors, autoimmunity, and infectious diseases:
“Solid Tumors - The scientific community has identified, robustly characterized and clinically evaluated over 75 specific antigen targets across multiple solid tumor types, and we plan to use this data to inform our next wave of product development in oncology. We intend to identify a “basket” of solid tumors that share a common set of highly immunogenic and clinically validated tumor-relevant antigen targets for inclusion in our next AIM ACT clinical program. We envision this program will evaluate a new AIM ACT product candidate as both monotherapy and in combination with a tumor microenvironment, or TME, altering therapy, such as checkpoint inhibitors. We plan to evaluate clinical POC to support the AIM technology in solid tumors, which would serve as the basis for the introduction of an AIM INJ product candidate into early clinical development for solid tumor indications”
“Autoimmune Disorders - We believe that our AIM technology will enable us to target autoimmune conditions using either the AIM ACT or AIM INJ modality. For most autoimmune disorders like Type 1 Diabetes, autoreactive (or self-destructive) T cells become the cells targeted for therapeutic intervention. For these conditions, AIM nanoparticles are loaded with Signal 1 antigen peptides that autoreactive T cells recognize, and Signal 2 is programmed to deliver a suppressive or apoptotic signal that either tolerizes or eliminates the disease-causing T cells. In conditions like multiple sclerosis, or MS, the Epstein-Barr virus, or EBV, plays a critical role in mediating the disease process. Eliminating EBV-infected cells with EBV-specific T cells has been shown by others to impact disease progression for patients with Primary Progressive MS. We believe that EBV-specific AIM ACT or AIM INJ product candidates can be developed for clinical evaluation in various forms of MS.
“Infectious Diseases - We believe that there may be significant opportunities to address other viral-mediated diseases using the AIM platform to develop either AIM ACT or AIM INJ product candidates. We also believe that the AIM technology may offer a novel approach to the rapid treatment of, and preparation for, future viral-epidemics and pandemics.”
Business model
The company’s business is a platform creating a pipeline of drug candidates. Neximmune is positioning themselves to use AIM to generate templates for immunotherapy combination treatments and off-the-shelf T-cell products in cancer and other diseases:
“Advance NEXI-001 and NEXI-002 to registrational trials. Our initial focus is on developing therapies for the treatment of hematologic malignancies where there are existing clinical and regulatory precedents as well as a broad clinical and preclinical dataset for comparison of risk/benefit effectiveness. Our first two product candidates, NEXI-001 for patients with AML and NEXI-002 for patients with MM, are both targeted at diseases with a successful history of cell therapy research, late-stage clinical development and product registration by others. We believe this will facilitate our ability to understand the performance of our product candidates relative to other products targeting similar patient populations.”
“Expand AIM ACT into solid tumors. We are expanding our pipeline into solid tumor indications. We expect our first product candidate targeting solid tumors to use a non-proprietary set of antigen peptides commonly over-expressed on a set of solid tumors, based on our existing preclinical work in solid tumor models. Given the large number of potential antigen combinations in solid tumors, we expect licensing and partnerships to be a core element of our strategy as we establish the broader applicability of our AIM technology.”
“Accelerate development of our AIM INJ modality. We believe that one of the most significant advantages presented by our AIM technology is the potential for an injectable form of the AIM nanoparticle. We expect that a key step in developing this technology will be to leverage experience and insight from our AIM ACT tumor product candidates into an injectable modality, which would be available as an off-the-shelf immunotherapy.”
“Leverage partnerships to drive new product development in autoimmune disorders and infectious diseases. The AIM INJ modality will be constructed to deliver either “suppressive” or “apoptotic” co-stimulation signals directly to auto-reactive T cell populations, which is critical for addressing autoimmune disorders. In addition to autoimmune disorders, we believe that there may be significant opportunities to address virally-mediated infectious diseases via either the AIM ACT or AIM INJ modality. We also believe that the AIM technology may be applicable to the treatment of, and preparation for, future virally-mediated epidemics and pandemics. While we believe that our AIM technology platform is well-suited to address these new therapeutic opportunities, we expect that we would partner with experienced biopharmaceutical companies with deep capabilities in these areas to advance new therapies in these potential indications. While we intend to establish our own internal capabilities to develop and commercialize our product candidates, we will also explore strategic collaborations or partnerships that may accelerate our development timelines, broaden the therapeutic reach of our AIM technology platforms and maximize the full potential of both the AIM ACT and AIM INJ modalities.”
Valuation
Neximmune’s main value is in its two lead drug candidates: NEXI-001/002:
“We have completed non-clinical work to advance the AIM INJ modality towards a potential IND filing, including preparing appropriate IND-enabling experiments in support of a planned clinical program focusing on solid tumors. Subject to regulatory feedback and an IND filing, we anticipate a second clinical program that would target autoimmune disease and which would be the first AIM product candidate to suppress, rather than activate, T cell function. In support of this potential program, we have generated and published pre-clinical data in which observe that AIM nanoparticles to engaged and suppress auto-reactive T cells.”
“Additionally, we are developing the AIM platform for potential clinic application in patients suffering from specific infectious disease. In non-clinical studies, we have been able to expand CD8+ T cells directed against viral antigens including Epstein-Barr virus (EBV), CMV, and HPV.”
“Moving forward, we expect to pursue additional modality applications and indications across multiple disease areas. Our strategy is to establish clinical proof of concept, or POC, for the AIM technology with NEXI-001 and NEXI-002 in hematologic malignancies, and then to develop new AIM ACT and AIM INJ product candidates to expand into solid tumors, with potential further expansion into autoimmune disorders and infectious diseases. We have generated a significant body of non-clinical data to support this approach, which we use to prioritize our clinical development efforts and to identify potential disease areas and indications to pursue.”
Mention highlights in the S-1
AIM: 237
Manufacturing: 186
Nanoparticles: 71
Antigen: 65
Biologics: 32
Immunotherapy: 31
AML: 25
Myeloid: 9