Axial partners with great founders and inventors. We invest in early-stage life sciences companies such as Appia Bio, Seranova Bio, Delix Therapeutics, Simcha Therapeutics, among others often when they are no more than an idea. We are fanatical about helping the rare inventor who is compelled to build their own enduring business. If you or someone you know has a great idea or company in life sciences, Axial would be excited to get to know you and possibly invest in your vision and company . We are excited to be in business with you - email us at info@axialvc.com
Gracell Biotechnologies is a cell therapy company based in China. Their recent S-1 highlights the company’s focus on CAR-T cell therapies, their FasTCAR and TruUCAR platforms, and the challenges/opportunities in the field. On the platform side, Gracell’s (communicated) advantages are:
FasTCAR - focused on developing T-cells with lower exhaustion profiles for autologous cell therapies along with next-day manufacturing (versus 2-6 weeks). Data in relapsed/refractory multiple myeloma in an ongoing Phase 1 trial in China.
TruUCAR - focused on developing T-cells that are non-HLA matched for allogeneic cell therapies and off-the-shelf use. Data in relapsed/refractory T-cell acute lymphoblastic leukemia (ALL) in an ongoing Phase 1 trial in China.
With these technologies the company is building a pipeline of autologous and allogeneic cell therapies in liquid and solid tumors. Gracell’s lead drug candidate, GC012F, comes from their FasTCAR platform and is focused on r/r multiple myeloma. As of July 2020, 15/16 patients responded (ORR=93.8%) with 6 patients taking the highest dose experiencing a complete response. GC027 is the lead candidate from the TruUCAR platform for r/r T-cell ALL with a 100% complete response rate so far.
Highlights
Gracell’s pipeline is focused on hematologic malignancies and solid tumors. They focus on starting trials in China with the long-term goal to run global trials and rely on in-house manufacturing.
Gracell is pursuing the cell therapy marketing more specifically chimeric antigen receptor T cells, or CAR-T cells. With a pipeline of autologous and allogeneic drug candidates, the company is placing bets on both technologies. Allogeneic might be more useful for hematological malignancies and autologous for solid tumors, and Gracell is developing next-generation products to build off the success of Kymriah (Novartis) for pediatric B cell acute lymphoblastic leukemia and Yescarta (Kite Pharma/Gilead) for diffuse large B cell lymphoma.
Gracell is building a vertically integrated cell therapy company using their technology platform to discover and develop new cell therapies and building out manufacturing capabilities to scale them up. Overtime, their pipeline will move into solid tumors and they will expand their >100K square feet facilities in Shanghai. Gracell’s current facilities support annual production of 3,200 autologous samples from the FasTCAR platform and 12,000 allogeneic samples from the TruUCAR platform.
Team
Gracell’s Founder and CEO is William Wei. Previously, he was the CEO of Cellular Biomedicine Group and worked at Chiron and Affymetrix. The company’s CMO, Martin Sersch, was the CMO at another CAR-T company, Mustang Bio and worked as a medical director at Amgen.
Investors
Gracell is based in China and therefore must follow PRC laws around foreign ownership. Therefore, they use an off-shore (Cayman Islands) holding company to own the work done in China but also a wholly foreign-owned enterprise in the US that runs R&D.
The company is founder-driven with Wei owning a little over a third of the company (through his trust based in the British Virgin Islands. OrbiMed among other venture capital firms are also investors.
Technology
Gracell’s technology is centered around FasTCAR and TruUCAR.
From the S-1:
“FasTCAR is designed to address the most pressing challenges associated with autologous therapies, such as lengthy manufacturing time, suboptimal manufacturing quality, high therapy cost and poor T cell fitness. We transform the three primary production steps—activation, transduction and expansion—into a single “concurrent activation-transduction” step. This is achieved by utilizing XLenti vectors derived from lentivirus to concurrently activate and transduce resting T cells and enable them to stably express one or more CARs and proliferate actively in vivo. In addition, FasTCAR manufactured CAR-T cells are younger, less exhausted and show enhanced proliferation, tissue migration and tumor cell clearance activities as demonstrated in preclinical studies, eliminating the need for the ex vivo expansion phase in the conventional process. This streamlined process significantly shortens the production time from an industry norm of two to six weeks and achieves next-day manufacturing. Shorter manufacturing time is of particular importance to increasing the widespread utility of CAR-T cell therapies, particularly in the case of rapidly progressing cancers. We established fully-closed production lines designed to produce FasTCAR product candidates while reducing the risk of contamination and optimizing cost-efficiency. Our significantly shorter manufacturing time and highly efficient manufacturing process may result in meaningful cost savings, increasing the accessibility of cell therapies for cancer patients. We are developing our lead autologous product candidate, GC012F, as well as multiple autologous clinical-stage pipeline candidates on our FasTCAR platform.
[and]
TruUCAR is designed to generate high-quality allogeneic CAR-T cell therapies that can be administered “off-the-shelf” at lower cost. As with FasTCAR, TruUCAR uses a lentivirus to deliver its CAR. TruUCAR has several key design differences when compared to conventional allogeneic CAR-T approaches. TruUCAR is designed to specifically target a patient’s T cells and natural killer, or NK, cells that would otherwise be directed against the foreign, or allogeneic, cells resulting in rejection by the patients. This feature allows our allogeneic cell therapies to survive a patient’s immune system without the need for combination treatment with anti-CD52 antibodies that may leave a patient at increased risk for infection. TruUCAR is designed to avoid GvHD, one of the most severe adverse events of allogeneic CAR-T cell therapies, and rapidly eliminate cancer cells without the need to bridge to hematopoietic stem cell transplantation, or HSCT, which is often used with conventional allogeneic CAR-T cell therapy to strengthen its therapeutic effects but pose a risk of early mortality. As a result, TruUCAR’s monotherapy approach has the potential to significantly reduce the cost and length of treatment by achieving fast remission and avoiding anti-CD52 treatment and potentially HSCT. We believe that TruUCAR may result in meaningful cost savings, further increasing the accessibility of cell therapies for cancer patients. We are developing our lead allogeneic product candidate, GC027, as well as multiple allogeneic pipeline candidates on our TruUCAR platform.”
Moreover, the company is designing new CAR constructs to reduce GvHD, cytokine signalling, antigen escape, engineer immunosuppressive tumor microenvironment (TME) and other things to improve patient responses.
Gracell’s pipeline is focused on hematologic malignancies and solid tumors. They focus on starting trials in China with the long-term goal to run global trials and rely on in-house manufacturing.
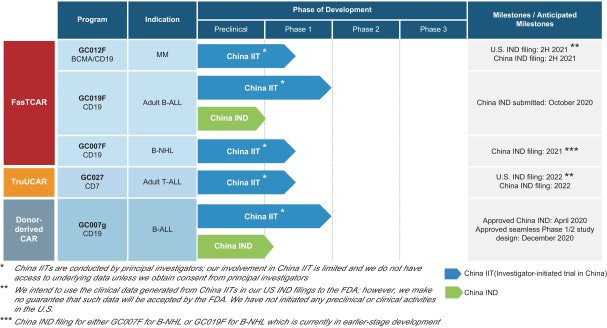
The company’s lead candidates are:
GC012F - “GC012F is a FasTCAR-enabled dual BCMA- and CD19-directed autologous CAR-T product candidate being studied for the treatment of MM in an ongoing investigator-initiated Phase 1 trial across multiple centers in China. As of July 2020, 16 r/r MM patients were enrolled and treated with 93.8% of these patients having high-risk features, which represent a subgroup of MM patients with a poor prognosis and potentially rapid disease progression, making them particularly challenging to treat even with novel agents. All patients in the trial had relapsed from, or were refractory to, previous treatments, including the most commonly used agents and SOC treatments. 15 of 16 patients achieved and maintained a response. In the highest dose cohort which is the recommended dosage level, 100% of the six evaluable patients achieved MRD- sCR as best response which was maintained through the landmark analysis at six months after CAR-T infusion. Based on these results, we expect to submit IND applications for GC012F in r/r MM to the FDA and the NMPA by the end of 2021.”
GC019F - “GC019F is a FasTCAR-enabled CD19-directed autologous CAR-T product candidate that has been studied for the treatment of adult B-ALL in a completed investigator-initiated Phase 1 trial across multiple centers in China. We submitted an IND application to study GC019F in B-ALL to the NMPA in October 2020, which was accepted by the Center for Drug Evaluation of the NMPA, or CDE. An investigator-initiated trial for GC019F for the treatment of r/r B-NHL is currently in the planning stage and is expected to begin patient enrollment by the end of 2020.”
GC007F - “GC007F is a FasTCAR-enabled CD19-directed autologous CAR-T product candidate being studied for the treatment of B-NHL in an ongoing investigator-initiated Phase 1 trial across multiple centers in China. Based on the clinical results from the investigator-initiated trial, we plan to submit an IND application for either GC019F or GC007 in r/r B-NHL to the NMPA in 2021.”
GC027 - “GC027 is a TruUCAR-enabled CD7-directed allogeneic CAR-T product candidate being studied for the treatment of adult T-ALL in an ongoing investigator-initiated Phase 1 trial across multiple centers in China. As of February 2020, five adult r/r T-ALL patients were enrolled and treated. All patients enrolled had relapsed from, or were refractory to, their prior line of therapy. All five evaluable patients achieved a CR or CRi, resulting in an ORR of 100%, including four patients, or 80%, achieving MRD- CR on Day 28 after treatment. CRS was observed in all patients and was resolved with treatment. No patient developed neurotoxicity or GvHD. We expect to submit an IND application for GC027 in adult r/r T-ALL to the FDA and the NMPA in 2022.”
GC007g - “GC007g is a donor-derived CD19-directed allogeneic CAR-T cell therapy that has been studied for the treatment of r/r B-ALL in a completed investigator-initiated Phase 1 trial, where CAR-T cells were manufactured using T cells from an HLA-matched healthy donor. We obtained IND approval to study GC007g in B-ALL from the NMPA on April 1, 2020, and were granted approval from the NMPA on December 24, 2020 for a seamless Phase 1/2 registrational trial. Our goal is to submit a biologics license application, or BLA, to the NMPA for GC007g upon completion of a registrational trial.”
Market
Gracell is pursuing the cell therapy marketing more specifically chimeric antigen receptor T cells, or CAR-T cells. With a pipeline of autologous and allogeneic drug candidates, the company is placing bets on both technologies. Allogeneic might be more useful for hematological malignancies and autologous for solid tumors, and Gracell is developing next-generation products to build off the success of Kymriah (Novartis) for pediatric B cell acute lymphoblastic leukemia and Yescarta (Kite Pharma/Gilead) for diffuse large B cell lymphoma.
Issues/opportunities are:
“Autologous cell therapies are highly personalized, making the manufacturing process time-consuming, complex, costly and difficult to scale. It is also challenging to generate sufficient high-quality T cells as T cells of patients are often compromised from earlier lines of cancer treatment.”
“Unlike autologous therapies that derive cells from patients, allogeneic therapies, including those intended for use off-the-shelf, derive cells from healthy donors but require modifications to reduce or eliminate host versus graft rejection, or HvG, where a patient’s immune cells recognize infused non-HLA-matched donor cells as foreign and reject them, and GvHD.”
“Additionally, despite progress in treating hematologic malignancies, CAR-T cell therapies have had little success with treating solid tumors, primarily as a result of CAR-T cells’ limited ability to penetrate and persist in solid tumors.”
The company also communicates 7 current limitations that Gracell can work on and build moats around:
“Manufacturing Time - Industry norm of two to six weeks. Patients must wait for a few weeks to be treated with their engineered cells, primarily due to the lengthy manufacturing time of two to six weeks, which is the current industry norm. Lengthy manufacturing time can prove suboptimal for those patients with rapidly progressing cancer who may die while waiting for the therapy.”
“Production Quality - High failure risks. The complex, multistep process of generating autologous CAR T cells increases the risk of manufacturing failure, including failure to generate a sufficient density of viable T cells or batch failures resulting from infection or contamination during production. Reported failure rates of autologous CAR-T cell manufacturing range from 5% to 14%. Manufacturing failure results in an inability to provide therapy.”
“Cost / Access - High manufacturing cost and lengthy hospitalization. High manufacturing cost and highly personalized nature of CAR-T cell therapies result in an average cost of US$1.5 million per patient per therapy. The long manufacturing time for conventional CAR-T cell therapies results in lengthy hospitalization that requires a dedicated infrastructure for in-patient care from specialized medical centers, further increasing costs to patients.”
“Poor T Cell Fitness - Exhausted T cells. T cells of patients used for the autologous CAR-T therapy are often compromised from earlier lines of treatment, resulting in decreased survival, proliferation, differentiation, homing and tumor killing ability. The T cells are further weakened during the activation and expansion phases in conventional CAR-T manufacturing processes, affecting the quality of CAR-T cells.”
“Limited Durability - Need for combination therapy. It is challenging to maintain response in relapsed or refractory patients for various reasons. For example, infused allogeneic CAR-T cells may be rejected by a patient’s immune system via HvG, which harms the durability and efficacy of the treatment. Anti-CD52 therapies are often co-administered to avoid HvG. Additionally, CAR-T cell therapies are often coupled with or bridged into HSCT to strengthen the therapeutic effects and improve response durability.”
“Relapse - Antigen escape. CAR-T cell therapies targeting a single antigen have been shown to lose efficacy due to antigen escape, which occurs when expression of a CAR-T target on a malignant cell is lost or reduced, resulting in an expansion of the malignant cells that have escaped the ability of the CAR-T cells to kill them. Antigen escape poses a significant risk of failure for CAR-T cell therapy and may result in response rates declining from the initial response level.”
“Limited Efficacy - Solid tumors. Despite progress in the treatment of blood cancers with CAR-T cells, achieving success in solid tumors is significantly more challenging due to a variety of factors, including inefficient trafficking of CAR-T cells to tumor sites, immunosuppressive TME, limited ability of CAR-T cells to penetrate and remain alive in solid tumors, target antigen heterogeneity, and the inability of ex vivo expanded CAR-T cells to persist and proliferate following infusion into patients.”
Business model
Gracell is building a vertically integrated cell therapy company using their technology platform to discover and develop new cell therapies and building out manufacturing capabilities to scale them up. Overtime, their pipeline will move into solid tumors and they will expand their >100K square feet facilities in Shanghai. Gracell’s current facilities support annual production of 3,200 autologous samples from the FasTCAR platform and 12,000 allogeneic samples from the TruUCAR platform.
Valuation
The company’s value is mainly driven by the clinical success of their pipeline, which is still in the early stages of development:
“We have generated a pipeline of autologous and allogeneic cell therapy candidates with the potential to treat both hematologic malignancies and solid tumors. Our clinical development strategy is built on the robust pre-IND investigator-initiated trials program that we have established in partnership with top-tier hospitals in China. We engineer, produce and provide CAR-T cells to the principal investigators at those hospitals for administration in patients. The principal investigators agree to provide us results and findings generated from the investigator-initiated trials. We do not have access to the underlying data points from these studies unless separately requested by us and approved by them. To the extent that, after discussions with the FDA and/or the NMPA, we are permitted to rely on all or part of these initial results and the underlying data points to support our regulatory filings with the FDA and/or the NMPA, we work in close collaboration with the principal investigators to collect the data with their approval. This strategy is designed to expedite our global clinical development activities with the initial results in investigator-initiated Phase 1 trials utilizing safety as primary endpoint and overall response rate, or ORR, as secondary endpoint. There is no guarantee that this strategy will be successful or will speed up the development of our product candidates. We have generated all our product candidates internally.”
Mention highlights in the S-1
PRC: 530
CAR-T: 327
Cell: 324
TruUCAR: 67
Health: 66
Invention: 20
CD19: 20
Freedom: 1