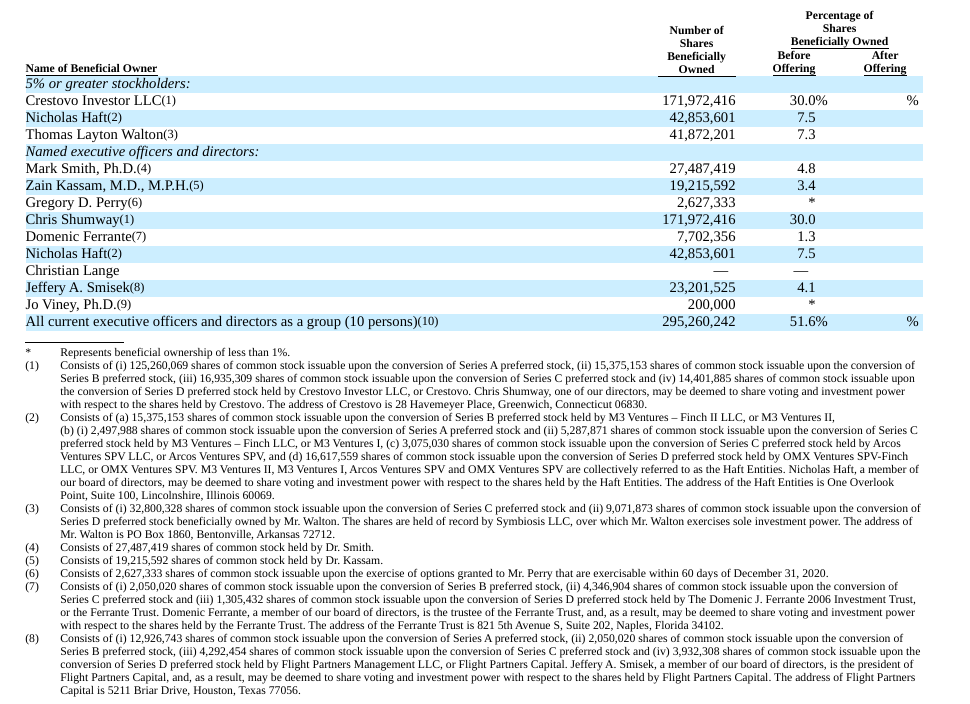
Axial partners with great founders and inventors. We invest in early-stage life sciences companies such as Appia Bio, Seranova Bio, Delix Therapeutics, Simcha Therapeutics, among others often when they are no more than an idea. We are fanatical about helping the rare inventor who is compelled to build their own enduring business. If you or someone you know has a great idea or company in life sciences, Axial would be excited to get to know you and possibly invest in your vision and company . We are excited to be in business with you - email us at info@axialvc.com
Finch Therapeutics recently filed an IPO to raise $100M to scale their microbiome drug development platform: S-1. The company develops microbiome therapeutics with a focus on gastrointestinal (GI) diseases. Finch’s Human-First Discovery platform relies on forward genetics to discover microbial cocktails to treat dysbiosis. Their technology relies on identifying microbes that are missing in a disease and designing therapeutics to replace them.
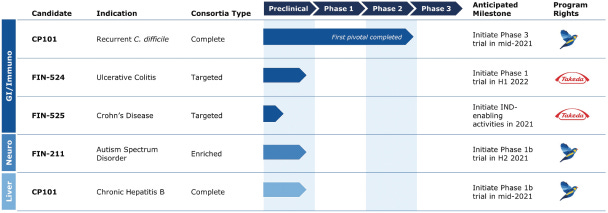
The company’s lead drug candidate, CP101, treats patients with recurrent Clostridioides difficile infection (C. diff). Finch is bringing this asset to a phase 3 clinical trial in mid-2021 along with developing other assets across diseases like chronic hepatitis B virus, autism spectrum disorder, and ulcerative colitis.
Highlights
The premise of Finch is that real-world data exists in healthy patient microbiomes to inform the development of new medicines. The company’s lead drug candidate, CP101, is a microbial community of microbes from healthy donors and formulated for oral delivery to treat C. diff.
This reverse translation approach gives Finch a strong ability to establish causal relationships within the human microbiome that may not be accessible in preclinical models. As Finch builds their own database of FMTs, the company will be in a stronger position to translate clinical data into new drugs. The company has strategic partnerships with OpenBiome (50K treatments) along with access to a library with more than 10K microbiome samples.
Ultimately, over 40B doses of antibiotics are used per year. This creates a massive opportunity for Finch to explore and understand where microbial function can be restored and for which diseases.
Team
Finch is “led by an energetic team of experienced biotechnology executives and recognized leaders in the microbiome therapeutics space.” The Co-Founder and CEO, Mark Smith, founded OpenBiome, a nonprofit for microbiota transplants, and earned his PhD at MIT in the Alm Lab. Smith started OpenBiome in 2012 after a family member got C. diff, which motivated the founding of the now largest stool bank in the world. This led to Finch’s formation in 2014 to develop microbiome drugs that mimic the formulation of stool samples from known, successful interventions.
Gregory Perry is Finch’s CFO and previously was CFO at Novelion Therapeutics, Aegerion Pharmaceuticals, and Eleven Biotherapeutics. The company’s Chief Medical Officer is Zain Kassam, also a Co-Founder, who was previously CMO at OpenBiome. Joseph Vittiglio is Finch’s General Counsel having worked at AMAG Pharmaceuticals, Flexion Therapeutics, and AVEO Pharmaceuticals.
Investors
Congrats Nick.
Technology
The premise of Finch is that real-world data exists in healthy patient microbiomes to inform the development of new medicines. The company’s lead drug candidate, CP101, is a microbial community of microbes from healthy donors and formulated for oral delivery to treat C. diff:
“In June 2020, we announced that CP101 met its primary efficacy endpoint in PRISM3, a randomized, placebo-controlled, multi-center, pivotal, Phase 2 clinical trial in recurrent CDI. Overall, 74.5% of participants who received a single administration of CP101 achieved a sustained clinical cure, defined as the absence of CDI through week 8, achieving statistical significance for the primary efficacy endpoint, with a clinically meaningful 33.8% relative risk reduction for CDI recurrence compared to placebo. In PRISM3, the prevalence of adverse events was similar across CP101 and placebo arms, with no treatment-related serious adverse events, or SAEs, in the CP101 arm. We plan to initiate a Phase 3 clinical trial as our second pivotal trial of CP101 for recurrent CDI in mid-2021 to build on the results of PRISM3. Further, based on the clinical validation of CP101 for recurrent CDI, we plan to develop CP101 in other diseases of dysbiosis, including the treatment of chronic HBV. We plan to initiate our first clinical trial of CP101 in chronic HBV in mid-2021, with an initial safety review in the second half of 2021 and topline data in the second half of 2022.”
“In addition to developing CP101, a Complete Consortia product candidate designed to address community-level dysbiosis, or disruption across many functional pathways and species, we are also developing Targeted Consortia product candidates that consist of individual bacteria grown from master cell banks to engage narrower pathway-level dysbiosis. The ability to pursue both of these product strategies enables us to tailor our product candidates to the pathophysiology of each indication. This combination of capabilities also enables us to pursue a third product strategy, Enriched Consortia, which addresses dysbiosis at both the community and pathway level.”
Finch’s “Human-First Discovery platform” relies on clinical interventional data. Fecal microbiota transplantation (FMT) has been used to treat over 50K patients across a wide range of diseases. Finch uses this real-world data to:
Identify diseases where addressing dysbiosis provides therapeutic benefit
Reveal the mechanisms that underlie these results
Uncover key microbes and functional pathways that drive these clinical outcomes
This reverse translation approach gives Finch a strong ability to establish causal relationships within the human microbiome that may not be accessible in preclinical models. As Finch builds their own database of FMTs, the company will be in a stronger position to translate clinical data into new drugs. The company has strategic partnerships with OpenBiome (50K treatments) along with access to a library with more than 10K microbiome samples:
“We have used our Human-First Discovery platform to develop FIN-211, an Enriched Consortia product candidate that we are advancing for the treatment of the gastrointestinal and behavioral symptoms of ASD. Scientific research in human and animal models have highlighted the “gut-brain axis” linking dysbiosis to neurological and neurobehavioral conditions, as the microbiome impacts the enteric nervous system and the production of neurotransmitters. This basic research is supported by a growing body of third-party clinical research. In an open-label, proof-of-concept FMT trial conducted by one of our collaborators, it was observed that, two years after treatment, 33% of the study participants who had previously been diagnosed with ASD were below the ASD diagnostic cutoff score for the Childhood Autism Rating Scale (CARS), a commonly used ASD diagnostic tool. Additionally, in a third-party, open-label randomized, controlled trial, children with ASD receiving FMT and behavioral therapy showed a statistically significant improvement in their behavioral symptoms compared to those receiving behavioral therapy alone. Both studies also observed marked improvements in the gastrointestinal symptoms that many autistic children suffer from. There are no FDA-approved therapies for the core symptoms of ASD and the total financial burden of care for this condition is estimated to exceed $100 billion in the United States annually. We have received feedback from the FDA that demonstrating a benefit for either gastrointestinal or behavioral symptoms of ASD could support a biologics license application. Building on our discussions with the FDA, we aim to continue to validate behavioral instruments as part of our clinical development plans. We have designed FIN-211 to address both aspects of ASD and plan to initiate a Phase 1 clinical trial of FIN-211 in ASD in the second half of 2021, with topline data in the second half of 2022. We believe FIN-211 has the potential to transform care for patients with ASD.”
“We are also advancing FIN-524 and FIN-525 as Targeted Consortia product candidates for the treatment of ulcerative colitis and Crohn’s disease, the most common types of inflammatory bowel diseases, or IBD. We are partnering with Millennium Pharmaceuticals, Inc., or Takeda, a wholly-owned subsidiary of Takeda Pharmaceutical Limited, to develop these assets. FIN-524 was discovered through the computational and molecular analysis of data from 147 patients treated with FMT and 19 observational studies of an additional 2,210 patients. We plan to initiate our first clinical trial of FIN-524 in ulcerative colitis in the first half of 2022. In addition, we are conducting initial discovery efforts on FIN-525, and pending Takeda’s review, we could initiate IND-enabling studies for FIN-525 in Crohn’s disease in the second half of 2021.”
“Our Human-First Discovery platform leverages clinical data to significantly reduce drug development time and translational risk. Given the distinct biology of the human microbiome, developing products by relying on laboratory and animal models alone is challenging. However, with our Human-First Discovery Platform, we have deployed powerful machine learning capabilities to integrate our proprietary FMT data with information from our human strain library. We believe this strategy reduces translational risk, because we only commence programs where clinical data already exists, thereby limiting the risk that effects seen in the laboratory will not translate to the clinic. Further, in the many indications like chronic HBV where we believe a Complete Consortia product strategy is attractive, we are able to enter the clinic directly with CP101, avoiding the time, costs and translational risks associated with traditional preclinical development. We believe that this approach is enabled by the favorable tolerability profile we have observed to date with CP101.”
“We have differentiated capabilities to develop both complete and targeted microbiome therapeutics. We have product candidates that address the distinct types of dysbiosis that lead to microbiome-mediated diseases. We have an orally administered Complete Consortia product candidate, which we believe enables both a potential near-term commercial opportunity in recurrent CDI, if approved, and the ability to expand into new therapeutic areas linked to community-level dysbiosis. We are also developing Targeted Consortia and Enriched Consortia product candidates that engage selected biological pathways to address more specific functional defects. This combination of capabilities enables us to develop product candidates that address each of the distinct types of dysbiosis that lead to microbiome-mediated diseases.”
Finch’s platform has 3 main parts: (1) using “human data to identify promising clinical indications, microbial mechanisms and a consortia that engages these mechanisms”, (2) “IND-enabling activities, including bioprocess and formulation development, quality control and cGMP production, and (3) clinical development. Excitedly, data from the clinical development stage can be fed back into the first step:
“Clinical Indication Selection: We aim to de-risk development by targeting indications with known underlying dysbiosis, an understanding of relevant mechanistic pathways and, critically, data from FMT that provide proof-of-concept that a microbial intervention has the potential to positively impact clinically meaningful outcomes. We have exclusive access to certain data and samples from groups that we believe are the largest providers of FMT in the world, including OpenBiome, which has delivered more than 50,000 FMTs to over 1,000 clinical sites. With more than 300 third-party clinical studies evaluating FMT around the world, we are uniquely well positioned to leverage this trove of clinical data to identify promising new drug development opportunities. We believe that by requiring a foundation of clinical data prior to indication selection and program initiation, our programs are already significantly de-risked before we begin development.”
“Target Identification and Validation: We use translational assays and high-throughput sequencing to generate curated datasets from FMT studies, observational clinical studies, and sometimes preclinical models, for each target indication. We then use our expertise in microbial ecology enhanced by our proprietary machine learning tools to identify microbiome compositions and functions that are deficient in our target population and whose restoration is causally linked to improved outcomes. We believe that observational clinical studies and preclinical models are valuable for generating mechanistic hypotheses which can then be validated using interventional data from FMT. Taken together, these efforts provide molecular and microbial targets, specific metabolites or bacteria, that are linked to clinical outcomes.”
“Candidate Selection and Consortia Design: To engage these targets, we deliver designed microbial consortia. The capability to deliver both a complete microbiome and targeted microbes gives us the flexibility to engage a diversity of mechanisms and therefore develop treatments for a wide range of indications. In diseases characterized by community-level dysbiosis like CDI, we are able to deploy Complete Consortia product candidates like CP101. In diseases where we are able to target pathway-level dysbiosis like IBD, we are able to deploy Targeted Consortia product candidates like FIN-524. Importantly, we have obtained exclusive access to a library of more than 10,000 samples from certain donors that have each been administered to patients through FMT. We are able to cryo-revive and manufacture strains from these samples, enabling precise matching of the exact strain that was associated with clinical outcomes with FMT. We believe that this direct chain of custody from a clinical sample into a Targeted Consortia significantly reduces translational risk and is uniquely enabled by our proprietary partnerships. We are also able to engage both the Targeted and Complete Consortia product strategies in a single Enriched Consortia product candidate like FIN-211 for conditions like ASD that have both community- and pathway-level dysbiosis. We believe we are uniquely positioned to align product strategy with mechanism because of our capabilities to address community- or pathway-level dysbiosis.”
“Bioprocess and Formulation Development: We have developed proprietary methods for growing, harvesting, purifying, preserving and delivering microbiome consortia. Of particular note, our advanced lyophilization technology enables the preservation of a complete microbiome in a stable formulation with more than two years of stability at 2°–8°C and more than six months of stability at room temperature to accommodate excursions during delivery and administration. Furthermore, we have developed orally administered, targeted release technologies, enabling intestinal release that facilitates robust pharmacokinetics. We believe that our deep expertise in bioprocess and formulation development have, and will continue to, enable rapid development of differentiated products.”
“Quality Control and Product Safety: Unlike other product candidates in development, we have developed manufacturing processes that do not rely on non-specific biocides like ethanol to exclude potential pathogens. Instead, each of our product candidates leverages molecular screening technology to exclude potential pathogens and harmful antibiotic resistance or virulence elements. This technology enables us to exclude unwanted agents without compromising potentially beneficial microbes. In addition to these purity assays, we have also developed both culture-based and culture-independent measures of viability to provide consistent potency across lots.”
“cGMP Production: We have developed cGMP production capabilities as a strategic asset, internalizing key activities that we believe we are uniquely positioned to execute, while externalizing activities that can be completed by third parties, in order to maximize our capital efficiency. As an example of this strategy in action, we are developing cGMP production capabilities for CP101, an orally administered Complete Consortia product candidate. With nearly a decade of operational experience and know-how enabling our Complete Consortia manufacturing platform, we believe we are the only company in the world with the cGMP capabilities required to enable this manufacturing process. By contrast, we have worked closely with third parties for the production of certain Enriched Consortia and Targeted Consortia product candidates. For these product candidates, there are rapidly maturing providers able to leverage analogous experience with large scale fermentation, including the required capital equipment and infrastructure to enable cGMP manufacture of these product candidates.”
“Progression: We have developed a suite of customized pharmacokinetic and pharmacodynamic assays to maximize learning from our clinical programs to guide progression through clinical development. Our pharmacokinetic assays quantitatively assess the engraftment, or colonization in the intestine, of our consortia. Our pharmacodynamic assays measure the production of microbial metabolites and their downstream effects on the host.”
“Expansion: When we initiate clinical development of a new program, we aim not only to inform the progression of the specific program under evaluation, but also to inform expansion into other indications. As an example, having determined that we are able to engraft a diverse microbial community and effectively restore missing metabolic pathways with CP101 for recurrent CDI, we are now able to expand CP101 into other indications, such as chronic HBV, that are tied to community-level dysbiosis. Because this community-level dysbiosis is common to many microbiome-associated diseases, we believe this particular product strategy may have broad applications, such that clinical validation in one indication de-risks the development of other indications with similar characteristics.”
“Iteration: In addition to positioning our clinical development and translational medicine strategy to generate data that may inform expansion opportunities into new indications, we also believe that clinical data generated from the development of product candidates like FIN-211 will provide a rich pool of data that we can mine with our Human-First Discovery platform to inform follow-on product candidates in the same indication with even more favorable product attributes. In this way, our clinical development is designed to feed back into discovery, enabling iterative improvement and life cycle management as we establish franchises in new indications.”
Ultimately, over 40B doses of antibiotics are used per year. This creates a massive opportunity for Finch to explore and understand where microbial function can be restored and for which diseases.
Market
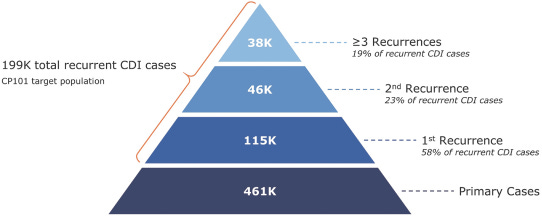
Finch’s lead market, C. diff, is involved in ~44K deaths per year in the US with an economic cost of $5B and 2.4M patient days. With over 200K cases of recurrent C. diff per year in the US and the standard-of-care (antibiotics) not restoring function, Finch along with other companies have the potential to make a large impact on these patients. The overall market opportunity Finch describes is the microbiome in general:
“The human microbiome describes the community of more than 30 trillion microbes that reside on and inside the human body. By evolving together over millions of years, microbes and humans have developed an intricate and mutually beneficial relationship that has only recently been uncovered. Enabled by the genomic revolution, researchers have discovered that humans carry over a 1,000-fold more microbial genes than host genes and that microbiome signaling is fundamentally intertwined with many aspects of human physiology ranging from immune and metabolic functions to neurological function and reproductive health. The deep inter-relationship between microbes and their human hosts is a co-evolution that has resulted in a learned dependency, leaving humans now reliant on inputs from this previously unrecognized organ system.”
“Disruption of the gut microbiome is associated with a large number of diseases that have dramatically increased in prevalence among populations in developed countries over the past century. We believe these epidemiological trends are linked to changes in the microbiome, which if reversed could potentially address an underlying cause of these diseases and change the epidemiology as a result. The rise of these chronic illnesses coincides with our adoption of a number of practices that disrupt the microbiome: more than 42 billion doses of antibiotics are administered annually, many killing 40-60% of microbial species in the gut; a third of babies in the United States today are born by caesarean sections, and are consequently unable to inherit this organ from their mother; and a highly sanitized and artificial environment, absent the environmental inputs expected by our microbiome, applies further pressure on this ecosystem within us. The effects of these environmental inputs coalesce around the gut microbiome resulting in dysbiosis and these changes are linked to a wide variety of chronic diseases. For example, antibiotic exposure doubles the risk of developing IBD, as well as significantly increases the risk of developing over 10 types of cancer. Early microbiome disruption is also associated with ASD, autoimmune indications such as celiac diseases, and allergies and asthma, and microbiome disruption later in life has been linked to neurodegenerative diseases, including Alzheimer’s disease and Parkinson’s disease. Importantly, in multiple animal models, these diseases can be induced by microbiome disruption and corrected by restoration, providing evidence of causality. For several of these therapeutic areas, this has been further bolstered by clinical data with FMT.”
“The effects of gut microbiome dysbiosis reverberates throughout the body, both because immune cells are heavily concentrated in the gut, where more than 70% of the body’s immune cells are located, and because microbial metabolites enter systemic circulation, acting on organs throughout the body. For example, researchers at the California Institute of Technology showed that the transfer of the microbiome from human donors with ASD into microbiome-free mice promoted hallmark autistic behaviors. In addition, a large body of research has documented the connection between over a dozen different microbiome species and molecular pathways connecting the gut’s enteric nervous system to the brain. We believe the gut-brain axis is but one example of how the microbiome can provide therapeutic benefits to diseases beyond the gut.”
“Restoring the microbiome, or its inputs, is an opportunity to directly address the underlying causes of many diseases driven by dysbiosis. Many existing drugs target only the downstream symptoms of disease, for example, anti-tumor necrosis factor, or anti-TNF, biologics are prescribed to IBD patients to suppress systemic immunity, without addressing the underlying drivers of gut inflammation and immune dysregulation. This can lead to unintended side effects as well as an incomplete resolution of disease. Treating the root cause of disease is more likely to deliver a therapeutic breakthrough and for many diseases of dysbiosis, we believe that only through the restoration of the critical physiological role of the microbiome organ can this be achieved. Currently there are no microbiome therapeutics approved by the FDA. We believe that our ability to target both community- and pathway-level dysbiosis through our Human-First Discovery platform uniquely positions us to deliver on this transformational opportunity to improve human health through microbiome therapeutics.”
The key value proposition of Finch’s C. diff. Drug is focusing on the first recurrence for patients and administering the drug in an outpatient setting. The latter could provide better pricing and reimbursement especially when compared to inpatient antibiotics.
Business model
Finch has built out a platform business with a diverse pipeline of microbiome drugs combined with a partnership for larger indications:
“Drive CP101 for recurrent CDI toward regulatory approval and commercialization. We achieved our primary efficacy endpoint, demonstrating superiority over standard of care alone in our first pivotal trial, while also observing no treatment-related SAEs. We plan to initiate a Phase 3 clinical trial of CP101 for recurrent CDI in mid-2021, with data expected in the first half of 2023.”
“Advance CP101 into additional indications where FMT demonstrates compelling clinical outcomes. With dozens of completed and hundreds of ongoing clinical studies by third parties with FMT, we have already identified several therapeutic areas where we believe addressing community-level dysbiosis through microbiome modulation has demonstrated compelling proof-of-concept clinical signals. We believe CP101 is uniquely well-positioned to translate these therapeutic insights into label expansion opportunities, without the translational risk associated with products that deliver only subsets of the full microbiome. We plan to initiate our first clinical trial of CP101 in chronic HBV in mid-2021, which will be the first example of this strategy in action.”
“Leverage our Enriched Consortia product strategy to drive clinical development of FIN-211 for the treatment of ASD and other high value indications. We intend to advance FIN-211, an Enriched Consortia product candidate for the treatment of ASD, into a Phase 1 clinical trial in the second half of 2021. We are initially focusing on pediatric ASD patients with gastrointestinal symptoms and see this patient segment as a natural bridge into the broader opportunities to engage the gut-brain axis, given the role of the microbiome in metabolizing and modulating important neurotransmitters.”
“Continue to use our Human-First Discovery platform to translate clinical data into a pipeline of differentiated product candidates, including Targeted Consortia. We plan to continue growing and evaluating our proprietary database and analytical tools to discover and develop new product candidates directly informed by clinical data. Importantly, because our machine learning algorithms become increasingly powerful as available data scales, we expect our efficiency to increase over time as we continue to expand our databases.”
“Selectively enter into strategic collaborations to maximize the value of our platform and pipeline. As the potential impact of this emerging modality becomes clear, we believe the breadth of our collaboration opportunities will expand, particularly since large pharmaceutical companies may focus on accessing technology from early leaders like us. While, if approved, we plan to independently commercialize our products in indications and geographies where we can maximize their value, given the breadth of our portfolio and the significant potential for this new modality, we intend to selectively enter into partnerships with biopharmaceutical companies whose capabilities and resources may accelerate the development of our pipeline. Our partnership with Takeda is an example of such a collaboration.”
Valuation
Most of Finch’s short-term value is tied up in its C. diff drug candidate, which is massively derisked by the recent Seres’ data and long-term, Finch's value is in its database of clinical samples to develop new medicines and garner other biopharma deals:
“Our commercial success depends in part on our ability to obtain and maintain proprietary protection for our current and future product candidates, novel discoveries, product development technologies and know-how; to operate without infringing on the proprietary rights of others; and to prevent others from infringing our proprietary rights. Our policy is to seek to protect our proprietary position by, among other methods, filing or in-licensing U.S. and foreign patents and patent applications related to our proprietary technology that are important to the development and implementation of our business. We seek to obtain domestic and international patent protection, and endeavor to promptly file patent applications for new commercially valuable inventions. We file new patent applications as we conduct research and development, initiate new programs, and monitor the activities of others within the microbiome field. We also rely on trademarks, trade secrets, know-how, and continuing technological innovation to develop and maintain our proprietary position.”
“The patent positions of biopharmaceutical companies like us are generally uncertain and involve complex legal, scientific and factual questions. In addition, the coverage claimed in a patent may be challenged in courts after issuance. Moreover, many jurisdictions permit third parties to challenge issued patents in administrative proceedings, which may result in further narrowing or even cancellation of patent claims. We cannot predict whether the patent applications we are currently pursuing will issue as patents in any particular jurisdiction or at all, whether the claims of any patent applications, should they issue, will cover our product candidates, or whether the claims of any issued patents will provide sufficient protection from competitors or otherwise provide any competitive advantage.”
“Because patent applications in the United States and certain other jurisdictions are maintained in secrecy for 18 months or potentially even longer, and because publication of discoveries in the scientific or patent literature often lags behind actual discoveries and patent application filings, we cannot be certain of the priority of inventions covered by pending patent applications. Accordingly, we may not have been the first to invent the subject matter disclosed in some of our patent applications or the first to file patent applications covering such subject matter.”
“Our patent portfolio leverages both offensive and defensive strategies to protect our business. We have a large and diverse patent portfolio consisting of more than 50 issued U.S. and foreign patents and more than 130 pending patent applications that we own or exclusively license from others. Our patent portfolio has broad applicability across the microbiome field, and provides protection for our lead product candidates CP101, FIN-211, FIN-524, FIN-525, as well as additional Complete, Enriched and Targeted Consortia product candidates that we may develop. For CP101 specifically, our patent portfolio includes more than ten U.S. patents that cover CP101 and methods of use and manufacture. These patents have expiration dates between 2031 and 2037.”
Mention highlights in the S-1
Patent: 234
Microbiome: 190
CDI: 146
Takeda: 94
Dysbiosis: 79
Co-Founder: 8
Formulation: 8
Library: 5