Axial partners with great founders and inventors. We invest in early-stage life sciences companies such as Appia Bio, Seranova Bio, Delix Therapeutics, Simcha Therapeutics, among others often when they are no more than an idea. We are fanatical about helping the rare inventor who is compelled to build their own enduring business. If you or someone you know has a great idea or company in life sciences, Axial would be excited to get to know you and possibly invest in your vision and company . We are excited to be in business with you - email us at info@axialvc.com
Bolt Biotherapeutics is a biotechnology company developing new immunotherapies for solid tumors that recently filed an S-1 to go public. The company is centered around its Boltbody ISAC technology - the platform relies on immunostimulants to engage and activate myeloid cells to kill cancer cells via phagocytosis and expose tumor neoantigens to the adaptive immune system. The premise of the technology is to convert cold tumors to hot ones where antigens are exposed to cytotoxic T-cells.
Bolt’s lead drug candidate, BDC-1001, is a HER2-targeting biosimilar (trastuzumab) conjugated to a TLR7/8 agonist to treat patients with HER2-expressing solid tumors (high and low). The drug candidate is in phase 1 trials, which is currently in the dose escalation part of the trial and is expected to go into phase 2 studies sometime in 2021. Bolt has established single agent anti-tumor activity of BCD-1001 in multiple preclinical models, including elimination of large tumors (~500 mm^3), as well as tumors that are refractory to trastuzumab or ado-trastuzumab emtansine. The company is also pointing it’s Boltbody ISAC platform to other targets such as CEA and PD-L1 with the former program expected to enter the clinic in 2022.
Highlights
The Boltbody ISAC platform focused on “activating and recruiting myeloid cells, thereby re-programming the tumor microenvironment to invoke an adaptive immune response.” To ensure that the drug candidates produced act locally even though delivery is done systemically, the platform focused on 3 parts, which they call “Three-Factor Authentication.”
The company’s Boltbody ISACs are designed with 3 primary components: (1) a tumor antigen-targeting antibody, (2) a linker that can be designed either as cleavable or non-cleavable, and (3) an immune stimulant to activate the patient’s innate and adaptive immune systems. These parts enabled Bolt’s biologics to activate for the innate and adaptive immune systems through different stages of the cancer immunity cycle to produce long-term anti-tumor activity.
Bolt’s main market is tumors that express HER2. HER2-targeting agents have only been approved for patients with HER2-positive breast and gastric cancers, with HER2-positivity based on protein overexpression or gene amplification. Only trastuzumab is approved for both indications. Additional approved HER2-targeting agents for HER2-positive breast cancer include the following: pertuzumab, trastuzumab emtansine, trastuzumab-hyaluronidase-oysk, lapatinib, neratinib, and most recently, trastuzumab-deruxtecan and tucatinib representing over $11B in annual revenues.
Team
The company was founded in 2015 based on the work from the Engleman Lab at Stanford. The lab was important for the approval of the first approved immunotherapy as well as inventing new technologies to activate dendritic cells in tumors in situ, without requiring their removal and activation in vitro, which is an important part of the Boltbody ISAC platform.
Bolt’s CEO is Randall Schatzman who was previously CEO of Alder BioPharmaceuticals. He was also a VP of Research at Celltech, director at Mercator Genetics, and section leader at Roche. David Dornan is Bolt’s CSO having previously been an SVP of Research and Manufacturing at the company and worked at Gilead and Genentech beforehand. Bolt’s CMO is Edith Perez. She was a VP at Genentech and worked at Mayo Clinic.
Investors
Bolt’s main investors at Novo and Vivo Capital. Edgar Engleman (inventor) amazingly still holds a little over 10% of the company. Bolt raised $173.7M in total on private markets from other firms as well such as Pivotal bioVenture Partners, Sofinnova Investments, Nan Fung Life Sciences, RA Capital Management, Surveyor Capital (Citadel), Rock Springs Capital, Pfizer Ventures, and Samsara BioCapital.
Technology
The Boltbody ISAC platform focused on “activating and recruiting myeloid cells, thereby re-programming the tumor microenvironment to invoke an adaptive immune response.” To ensure that the drug candidates produced act locally even though delivery is done systemically, the platform focused on 3 parts, which they call “Three-Factor Authentication:”
Tumor antigen recognition - selective and specific tumor-targeting Boltbody ISACs recognize and bind specifically to the target antigen-expressing tumors.
FcR-dependent phagocytosis - engagement of optimized Fc domains triggers myeloid-mediated phagocytosis of the Boltbody ISAC-bound tumor cell. This process directly kills antigen-expressing tumor cells and delivers tumor neoantigens to myeloid cells.
TLR-mediated activation - TLR agonist conjugates activate myeloid cells and enable the presentation of tumor-associated neoantigens to cytotoxic T cells, thereby initiating the body’s adaptive anti-tumor immune response and converting immunologically cold tumors to hot tumors. Furthermore, these activated myeloid cells also encourage additional myeloid cell-mediated phagocytosis to amplify the innate and adaptive immune responses.
During this “Three-Factor Authentication” process, tumor-associated myeloid cells engulf the Boltbody ISAC-bound tumor cells, become armed with tumor neoantigens, and migrate to the lymph nodes where they mediate the activation and rapid expansion of tumor-reactive T cells to eliminate tumor cells, including those without the initial target antigen. As a result, the patient’s immune system determines which neoantigens are most important to eliminate the target tumors. This enables antibodies that Bolt discovers to engage the immune system at different points in the cancer immunity cycle:
Ability to address difficult-to-treat solid tumors including those refractory to current treatments
Engaging the body’s innate and adaptive immune responses
Generation of immunological memory with epitope spreading to provide long-term anti-tumor responses and protect against recurrence
Ability to target tumor antigens with less dense cell surface expression
Capability to modulate myeloid cell activity via TLR potency and selectivity and Fc engineering
Well tolerated in preclinical studies by avoiding unintended systemic immune stimulation
Potential to benefit patients who have a defective adaptive immune response
The company’s Boltbody ISACs are designed with 3 primary components: (1) a tumor antigen-targeting antibody, (2) a linker that can be designed either as cleavable or non-cleavable, and (3) an immune stimulant to activate the patient’s innate and adaptive immune systems. These parts enabled Bolt’s biologics to activate for the innate and adaptive immune systems through different stages of the cancer immunity cycle to produce long-term anti-tumor activity.
Bolt’s pipeline has 3 major programs targeting HER2, CEA, and PD-L1. The company’s lead program, BDC-1001 is described below:
“Our lead product candidate, BDC-1001, is currently in clinical development for the treatment of patients with HER2-expressing solid tumors, including those with HER2-low tumors. BDC-1001 provides a compelling example of the potential of Boltbody ISACs to address unmet medical needs in solid tumors. BDC-1001 is delivered systemically and acts locally by targeting HER2-expressing tumors and related metastatic disease, triggering their destruction by the innate and adaptive immune systems. BDC-1001 consists of a biosimilar of the humanized monoclonal antibody trastuzumab that is chemically conjugated to one of our proprietary TLR7/8 agonists via a non-cleavable linker. We have observed through our preclinical studies that BDC-1001 is an activator of human myeloid antigen presenting cells that may kill tumors via three distinct mechanisms: trastuzumab-mediated cell killing, robust immune activation and induction of immunological memory. Our observed preclinical anti-tumor response coupled with a lack of adverse safety signals in our non-human primate GLP toxicology studies leads us to believe that BDC-1001 offers the potential for long-term and meaningful response for patients with HER2-expressing cancers, including certain HER2-low tumors. We initiated a Phase 1/2 trial of BDC-1001 in the first quarter of 2020 for the treatment of patients with HER2-expressing solid tumors. We are currently in the dose escalation portion of the trial and expect to move into Phase 2 dose expansions in 2021. We believe that our preliminary Phase 1/2 data provide us with clinical proof of concept for our HER2 Boltbody ISAC approach.
“Mechanism of Action - BDC-1001 stimulates anti-tumor activity with a three-pronged approach: direct tumor cell killing by trastuzumab-mediated mechanisms, localized phagocytosis and elimination of HER2-expressing tumor cells by activated myeloid APCs and durable immunity manifested by T cells reactive to tumor-associated antigens or neoantigens. These mechanisms are supported by our in vivo data demonstrating tumor elimination and immunological memory when treated with our BDC-1001 surrogates. The mechanism governing myeloid cell activation is tripartite with BDC-1001 binding to HER2-expressing tumor cells via the antibody variable region, leading to phagocytosis and tumor cell killing by myeloid APCs expressing Fcg receptors, or FcRs, such as macrophages, dendritic cells and monocytes. Once internalized, the TLR7/8 agonist attached to BDC-1001 gains access to the phagolysosome and mediates downstream events associated with TLR7/8 activation, including increased cytotoxicity, cytokine secretion, recruitment of immune effector cells and the processing and presentation of tumor-associated antigens that stimulate T cell-mediated immunity. Taken together, the downstream effects of myeloid APC activation induced by BDC-1001 results in the conversion of immunologically “cold” tumors into “hot” tumors. Activated myeloid APCs migrate to the draining lymph nodes following BDC-1001 mediated phagocytosis of HER2-expressing tumor cells. Upon arrival to the draining lymph nodes, activated APCs present the full diversity of potential tumor-associated antigens and neoantigens located within the phagocytosed tumor cells on peptide-MHC complexes to naïve and antigen experienced or previously exhausted T cells. This process, in conjunction with elevated co-stimulatory molecule expression following TLR7/8 recognition in myeloid APCs, leads to the polyclonal activation and expansion of T cells. As a result, the patients’ own immune system determines which are the relevant T cells to mobilize for tumor destruction and subsequent immunosurveillance, providing a compelling example of how an off-the-shelf targeted immunotherapeutic such as BDC-1001 can deliver a personalized therapeutic outcome.
Our lead product candidate, BDC-1001, is currently in clinical development for the treatment of patients with HER2-expressing solid tumors, including those with HER2-low tumors. We have designed BDC-1001 as a Boltbody ISAC comprised of a HER2-targeting biosimilar trastuzumab conjugated to one of our proprietary TLR7/8 agonists to maximize the potential anti-tumor response. Through our preclinical studies in mice, we have demonstrated that systemic administration of HER2 Boltbody ISACs exhibited localized immune activation that resulted in single agent activity that eliminated large (~500 mm3) tumors and generated immunological memory against cancers with epitope spreading. Furthermore, preclinical data showed anti-tumor activity against established tumors resistant to trastuzumab and ado-trastuzumab emtansine, and immunological memory providing protection against tumor cells that no longer express the HER2 antigen. Our observed preclinical anti-tumor response coupled with a lack of adverse safety signals in our non-human primate GLP toxicology studies leads us to believe that BDC-1001 offers the potential for long-term and meaningful response for patients with HER2-expressing cancers, including HER2-low tumors. We initiated a Phase 1/2 trial of BDC-1001 in the first quarter of 2020 for the treatment of patients with HER2-expressing solid tumors. We are currently in the dose escalation portion of the trial and expect to advance into Phase 2 dose expansions in 2021 in four clinically important and commercially compelling indications. We believe that our preliminary Phase 1/2 data provide us with clinical proof of concept for our HER2 Boltbody ISAC approach.”
Market
Bolt’s main market is tumors that express HER2. HER2-targeting agents have only been approved for patients with HER2-positive breast and gastric cancers, with HER2-positivity based on protein overexpression or gene amplification. Only trastuzumab is approved for both indications. Additional approved HER2-targeting agents for HER2-positive breast cancer include the following: pertuzumab, trastuzumab emtansine, trastuzumab-hyaluronidase-oysk, lapatinib, neratinib, and most recently, trastuzumab-deruxtecan and tucatinib representing over $11B in annual revenues.
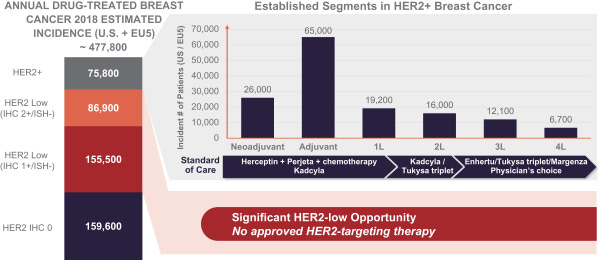
Incidence of breast cancer in the United States and EU is over 400K with over 75K HER2-positive. Out of the latter population, around half of patients have low HER2 expression that may not respond to other targeted therapies. This is Bolt’s first patient population they are focusing on.
“Trastuzumab-deruxtecan and tucatinib are important recently approved agents for the treatment of patients with previously treated advanced HER2-positive breast cancer. While both these agents provide important options for patients with advanced breast cancer, it is important to highlight the large percentage of patients who do not respond to these therapies or develop tumor progression after initial response. There are no approved treatments for either of these patient groups.
Despite the availability of these HER2-targeted agents, most patients with advanced disease and many with early disease are not cured and require multiple lines of therapy to achieve disease control, improve quality of life and extend survival. Additionally, there are patients not recognized in the current HER2-positive treatment paradigm such as those with lower HER2-expressing tumors or with HER2-expressing tumor types other than breast and gastric
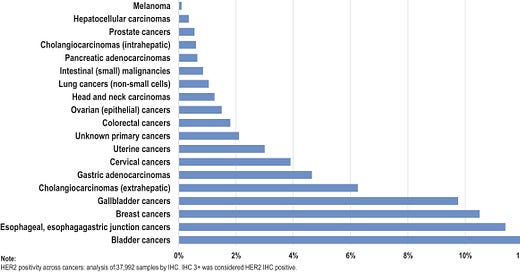
This unmet medical need includes patients with other tumor types, such as gastric cancer, NSCLC, CRC and bladder cancer, both for HER2-positive and HER2-low cancers. HER2 protein expression and overexpression have been well documented in a wide range of malignancies. Relative patient numbers for HER2 protein expression in these select tumor types are detailed in the figure below. This represents a large opportunity for a HER2 therapy utilizing our Boltbody ISAC approach.”
Business model
Bolt’s vision is to become a vertically integrated drug company centered around their Boltbody platform and expertise in myeloid biology:
Use the Boltbody ISAC approach and myeloid expertise to develop our pipeline of immune-activating therapies - “expertise in myeloid biology and immuno-oncology has led us to research various tumor antigens across solid tumors where significant unmet medical needs remain. Our expertise in medicinal chemistry and mAb engineering and our ability to modulate TLR linker-payloads allow us to optimize the therapeutic profile of our product candidates for any particular tumor antigen as part of our research and discovery efforts to produce durable anti-tumor responses. We believe that our approach is applicable to a broad spectrum of tumor-associated antigens expressed on cancers, including those that are refractory to existing therapies.”
Advance the development of BDC-1001, for the treatment of patients with HER2-expressing cancers - “BDC-1001 is currently in an ongoing Phase 1/2 clinical trial for the treatment of patients with HER2-expressing solid tumors. Based on our promising preclinical activity, BDC-1001 has the potential to be effective both as a monotherapy and in combination with existing therapies for patients with HER2-expressing solid tumors. While currently approved HER2-targeting agents are important and effective treatment options for some patients with HER2-expressing solid tumors, a large percentage of patients do not respond to these therapies, develop tumor progression after initial response or are not indicated for current HER2-targeting therapies. These sizable patient populations do not have adequate treatment options available to them. Therefore, we intend to rapidly advance development of BDC-1001 across multiple HER2-expressing cancers, including in both HER2-expressing and certain HER2-low cancers.”
Advance pipeline focused on additional promising targets including CEA and PD-L1 - ‘our robust pipeline includes BDC-2034 targeting CEA and a PD-L1 Boltbody program for which we have observed promising preclinical activity. These programs represent additional opportunities to differentiate our Boltbody ISAC approach from traditional immuno-oncology therapies that seek to inhibit key oncology pathways. By contrast, our Boltbody ISACs utilize target tumor antigens to bring nearby myeloid cells to the targeted tumor microenvironment to initiate robust innate and adaptive immune responses. We believe that this differentiated approach could improve the lives of patients by producing durable anti-tumor responses. We expect to designate our next clinical candidate in 2021.”
Invest in our myeloid expertise and Boltbody ISAC approach to explore the full potential of our targeted immunotherapies for the treatment of cancer - “our expertise, rigor and unbiased data-driven approach may lead to additional research and discovery programs that are complementary or independent of our Boltbody ISAC approach and our growing library of innate immune stimulators. Our research and discovery efforts are exploring additional immune agonists for the Boltbody ISAC approach as well as identifying novel targets in tumor-associated myeloid cells that can be targeted for anti-tumor outcomes. We believe such agents have the potential to reprogram tumor-supportive macrophages into tumor-destructive macrophages to elicit a productive anti-tumor immune response. This approach could potentially provide an avenue to further develop precision medicine with an immune modulator.”
Selectively enter into collaborations to expand Boltbody ISAC approach to future drug candidates - “in order to advance treatment options for patients, we may selectively collaborate with other companies with complementary technology or resources that could maximize the value of our product candidates and also expand our pipeline. Such collaborations may provide us with novel technologies, targets, agents or approaches that complement our myeloid expertise and innovative Boltbody ISAC approach to improve the lives of patients with cancer.”
Valuation
Bolt is mainly valued for its lead asset, BDC-1001, and pipeline. The company has used its platform and expertise to build up a pipeline of immune-stimulating, myeloid-engaging therapeutics.
Mention highlights in the S-1
Boltbody: 164
Stanford: 123
HER2: 52
Trastuzumab: 37
PD-L1: 34
TLR: 31
Lung: 12
Agonism: 6