Axial - New frontiers #2
New frontiers in life sciences
New frontiers #2
HSC therapies
Kidney disease
Royalty aggregators
Telemedicine
Machine learning and DELs
AAV manufacturing
iPSC-derived cell therapies
Voice in healthcare
N-of-1 trials
Patient-driven drug development
Axial partners with great founders and inventors. We invest in early-stage life sciences companies often when they are no more than an idea. We are fanatical about helping the rare inventor who is compelled to build their own enduring business. If you or someone you know has a great idea or company in life sciences, Axial would be excited to get to know you and possibly invest in your vision and company . We are excited to be in business with you - email us at info@axialvc.com
As always, any list or group of companies is not comprehensive; some companies are stealth-mode and it’s not appropriate to discuss them publicly and I don’t like spending too much time copying and pasting logos.
HSC therapies
Hematopoietic stem cells (HSC) are the source of blood cells from lymphocytes, red blood cells, monocytes among others. HSCs come in two lineages:
Lymphoid - encoding lymphocytes and NK cells
Myeloid - encoding red blood cells, macrophages, and more (this is a large opportunity to make more precise medicines for myeloid-derived diseases since this lineage encodes very essential cells that can lead to toxicity for myeloid-targeting medicines)
Over the last 6 decades, HSC transplants and medicines have become essential to cure several diseases from cancer to blood disorders. The first successful bone marrow derived HSC transplant was performed in the 1950s by E. Donnall Thomas at Fred Hutchinson Cancer Research Center; he won a Nobel Prize for this. Thomas infused bone marrow cells to repopulate the bone marrow of the patient to produce new blood cells. In 1961, the HSC was defined and characterized by two features: (1) self-renewal and (2) can produce different types of blood cells. Simply, resetting the immune system with an HSC transplant is incredibly powerful and curative. The procedure can be divided into two categories:
Allogeneic - hematopoietic stem and progenitor cells (HSPC) are sourced from a healthy donor and used to re-populate another patient’s hematopoietic and immune systems (~40% of transplants)
Autologous - the patient's own HSPC are used for a transplant (~60%)
To ensure the transplant engrafts and long-term durability for the patient, the material sourced must have HSCs. Finding healthy and immune compatible HSCs is another issue. As well as the conditioning regimens that are required to reset the immune system and improve the odds of a successful HSC engraftment. As a result, HSC transplants have pretty high mortality rates, which limits its uses in non-malignant/non-life-threatening diseases. However, if these issues can be solved, HSCs have the potential to create long-term cures for immune-related disease. For example, checkpoint inhibitors and CAR-T cell therapies have issues with response rates and sometimes safety. Combining these medicines with HSCs have the potential to lead to strong response rates with durable effects:
Source: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3901057/
However, HSCs face several barriers from becoming more widely used:
HSCs are unable to proliferate and differentiate in vitro. There is a high need for new sources of healthy and immune-compatible HSCs. Umbilical cord blood is a source of a large number of HSCs but has high costs and long timelines for collection, enrichment, HLA typing, and more.
Then determining how many HSCs are in a transplant is pretty difficult. Around 1 out of 10,000 cells in the bone marrow are thought to be HSCs with that number becoming 1 in 100,000 for blood. Developing better biomarkers (i.e. CD34) and sorting tools are needed.
HLA matching is important before a HSC transplant; however, there are low HLA matching rates in the human population. Finding ways to enable successful transplants without HLA matching is important or creating large registries to make matching easier. Figuring this out with minimizing/eliminating graft-versus-host-disease (GvHD) is important. GvHD is probably the biggest barrier for HSCs.
Multiple infusions are often required that can lead to infusion reactions, late toxicities, and relapse in patients.
Expanding and collecting HSCs; developing better harvesting methods and mobilization drugs is valuable.
Engineering HSCs with gene transfers (i.e. viral) and editing may expand the capabilities of an HSC. Ex vivo methods are limited by the small proportion of HSCs in a bone marrow population.
Conditioning regimens like chemotherapy and radiation are used to clear out a patient's blood system to improve the probability of an HSC transplant (i.e. prevent GvHD). Antibodies against targets like CD47 and CD45 have been shown to improve engraftment with less toxicity.
Ultimately, if these problems are solved, HSC medicines have the potential to cure, or at least improve patient mortality rates substantially, most diseases driven by the human immune system.
Kidney disease
Kidney disease affects over 30M people in the US and accounts for over $100M in annual healthcare costs. Without effective treatments, patients have to undergo procedures like dialysis and transplantation that lower a patient’s quality of life and lead to high healthcare costs. Excitedly, new tools combined with genomics has led to an improving understanding of kidney biology. Combined with new surrogate endpoints, drug development in kidney disease is set up to transform patient lives.
Companies like Goldfinch Bio, Chinook Therapeutics, Purespring Therapeutics, and Walden Biosciences are bringing new tools to develop medicines particularly for rare kidney diseases with defined clinical development milestones:
Genomics: several GWAS studies have been done to discover new genetic variants driving kidney diseases - targets, pathways, and MoAs - https://pubmed.ncbi.nlm.nih.gov/22143329/
Proteomics: kidney-generated proteins are useful signals of how the disease has progressed. For example, the CKD273 classifier, based on 273 urinary peptides, has been shown to be useful to detect chronic kidney disease (CKD) early - https://pubmed.ncbi.nlm.nih.gov/23690958/
Metabolomics: this is the toolkit that is theoretically most tractable given that there are ~10^3 metabolites versus ~10^4 genes and ~10^6 proteins. A few metabolites that are indicative of certain kidney diseases are γ-butyrobetaine, citrulline, and kynurenine - https://pubmed.ncbi.nlm.nih.gov/23052862/
Within kidney disease, the big opportunity is CKD. Current treatments only delay the onset of end-stage renal failure and have some really bad side effects. Around 20M in the US have CKD with 500K of them having end-stage versions, which requires dialysis or a transplant. For CKD, new MoAs to pursue are (inhibiting the renin–angiotensin–aldosterone pathway has been the traditional approach centered around blood pressure): loss of podocytes and renal epithelial cells, chemokines, JAK inhibitors, and ECM deposition. Who is building omics platform businesses to cure kidney disease?
Royalty aggregators
Drugs, and any product/company in general, can be viewed as a stream of cash flow over its lifetime. By having so much technical risk combined with IP monopolies if approved, drugs are a rich place to executive unique royalty deals: the drug developer can take some money off the table and the purchaser can acquire a potential lottery ticket winner with a set amount of investment. However, the market pricing mechanism is still pretty inefficient here. There is a massive opportunity to bring the software practices of Opendoor and Affirm to drug royalties. Is there anyone out there that is doing this or wants to?
Key parts of a royalty are:
The royalty rate and type - fixed versus tiered
Duration - how long the royalty agreement lasts?
Type of IP - is the royalty on a patent, trademark, or something else?
Product stage - preclinical, clinical, approved, on the market
Potential profitability - is your product pursuing a large and underserved market? You might be able to negotiate a lower royalty rate.
Exclusivity - is the royalty agreement exclusive for the use of IP? Just in case a similar product is developed; this is more downside protection.
Royalty Pharma is the market leader for late-stage and approved products. Ligand Pharmaceuticals for mid-stage products. And XOMA has emerged as a leader for early-stage products. There is also an opportunity to build a similar business model for preclinical products (this is a space where a company can help an inventor benefit from the work).
Telemedicine
Telemedicine has emerged as a standard way to deliver healthcare. Before COVID, the use of telemedicine was still relatively small when compared to the number of overall visits, and now, practices of all sizes rely on telemedicine to engage and monitor their patients. COVID was accelerated reimbursement for telemedicine, broken down some practice regulations that require an in-person consultation before telemedicine, and increasing data showing at least cost parity, and even cost savings, for telemedicine versus some forms of care.
Telemedicine is driven by network density with incumbent companies (i.e. Teladoc) and insurers (i.e. UnitedHealthcare) having the strongest advantages. These companies not only build telemedicine software products but also create a network of physicians. As a result, companies will have to develop products with telemedicine embedded or solve the problems telemedicine creates. The key theme is that what can be done with telemedicine, will:
Using telemedicine to scale a physician’s capabilities. The asynchronous nature of telemedicine allows a physicians to triage care and focus on patients with the highest need - Teladoc
To treat a patient - Virta Health, Livongo
Monitor a patient remotely (this is a large opportunity for process automation software) - Optimize Health, Gyant
Telemedicine will create new opportunities:
Increasing the acceptance of convenience products (i.e. Hims, The Pill Club)
Allowing home health to become more accepted. This is part of a larger move to deliver care outside of the hospital.
Develop better auditing tools for telemedicine
Building out integrations with digital devices to track a patient’s lifestyle. This can improve value-based care and improved clinical trials.
Delivering prescriptions and other products to the patient’s home
Asynchronous patient onboarding products (i.e pricing, eligibility, pulling medical records). Combined with this is billing and collections for telemedicine.
Ultimately, telemedicine companies will need integrated products with higher margins (i.e. treating a disease) and switching costs (i.e. FDA approval, clinical studies). A pure telemedicine product is commodified with the acquisition costs to build a new network prohibitively high. On the other side, a digital health product faces barriers to adoption that embedding within a telemedicine product can solve. Overtime, bundling several products into one can make costs savings for payors easier to predict and help the patient throughout their lifespan.
Machine learning and DELs
DNA-encoded libraries (DEL) are a tool to screen billions of chemical compounds. DELs tag each compound with a unique DNA barcode (~20-base-pair DNA sequences) to enable templated synthesis and tracking. The premise of DELs was driven by the difference between small molecules and biological molecules: the latter are produced by a genetic code and the former are not. Biological molecules can replicate and be selected for; whereas, small molecules don’t have these intrinsic features. The idea of a DEL started with the idea that adding a piece of DNA to a small molecule can imbue a code onto them and allowing the application of selective forces to identify small molecules with certain features:
Version 1: large libraries of small molecules with little-to-no directed evolution; companies like X-Chem, HitGen, and Vipergen; bottleneck was sequencing capabilities
Version 2: larger libraries using directed evolution; companies like DiCE, Ensemble (from the Liu Lab), and Nuevolution (now part of Amgen); sequencing became cheaper and more powerful
Version 3 will have to focus on diversity, medicinal chemistry, and relieving the bottlenecks from pooling and designing new selection methods; sequencing is pretty cheap with new tools like machine learning and microfluidics coming in. Machine learning will be particularly useful to map out SARs more rapidly. An exciting development is screening libraries against whole cell environments and membranes going beyond soluble protein targets.
In particular, DELs are becoming useful to train machine learning models for virtual screening. DELs can profile ~10^9 small molecules (versus 10^6 for high-throughput screening methods) while there are around 10^60 possible small molecules. As a result, virtual screening using machine learning can more efficiently map out structure-activity relationships (SAR) across this chemical space. So DELs are a cheaper way to generate the SAR data for models to have increasingly stronger predictivity. The physical screening of a DEL provides positive and negative hits for a model. If DELs can make virtual screening useful, developing a drug for a given target might add another step: (1) traditional screen, (2) DEL, (3) virtual screen.
Some problems to solve to ensure that DELs help build accurate virtual screening models are:
Improving the diversity of DELs - scaffolds and structural diversity of the chemical matter. A problem of using DELs to train machine learning models is that a class of compounds that bind a target might not be in the library to begin with. Ensuring that molecules in a DEL are sufficiently unique from one another is incredibly important. The issue is that sometimes the chemistry required to make certain chemical matter might not be able to get tagged by DNA.
Better filters for virtual screens: structural, removing duplicates and molecules with multiple reactive groups, predicting ease of synthesis
Expand a DEL screen beyond competitive binding of a target to select for more features like potential toxicity and ADME. This will feed into more accurate models and virtual screens.
AAV manufacturing
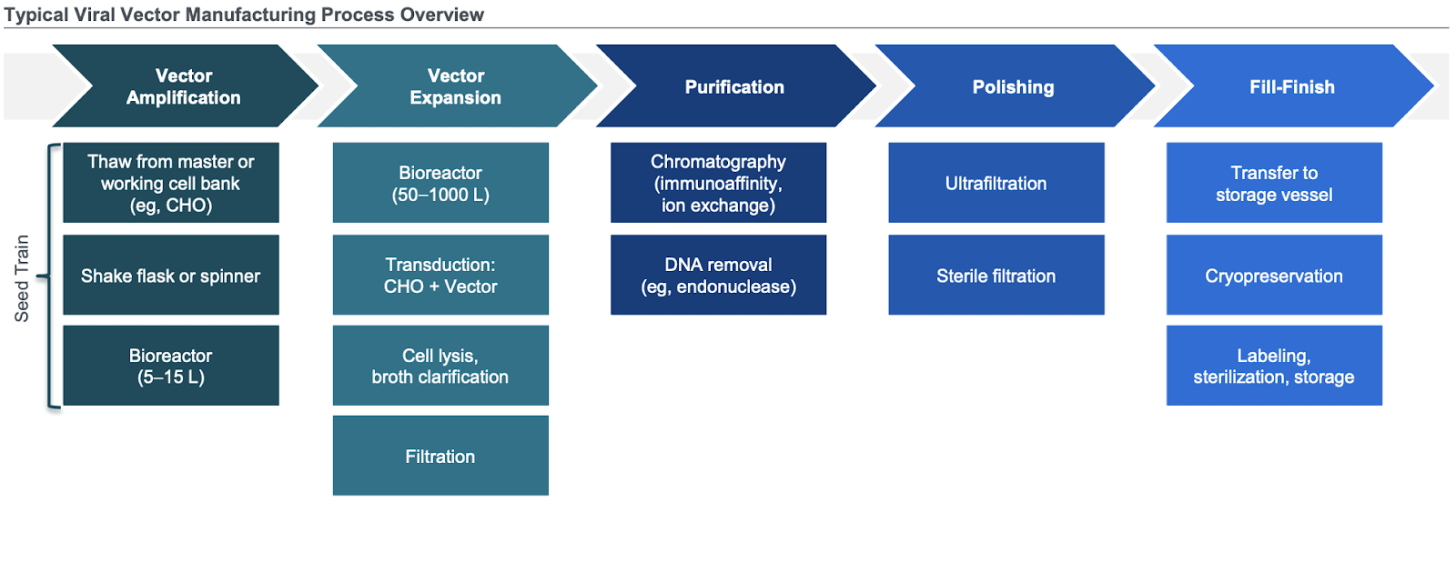
Biomanufacturing of adeno-associated virus (AAV) is composed of 5 main steps:
Cell culture (insect cells, HEK293) - HEK293 is the standard
Culture (suspension, 3D adherent, 2D) - suspension
Vector production (transient expression, stable cell line) - transient and takes 2-3 days
Recovery (filtration, centrifugation, TFF) - filtration and TFF
Purification (ultracentrifugation, chromatography) - chromatography
The first AAV gene therapy approved was Glybera in 2012 for LPLD in the EU followed by FDA approval of Luxturna in 2017 for IRD and Zolgensma in 2019 for SMA. As more of these medicines come through the clinic, more manufacturing capacity is required to expand to indications with larger patient populations.
Currently, two methods are the standard for AAV manufacturing - HEK293 and Sf9:
HEK293 cells that are adapted for suspension and are transfected with plasmid DNA. A master cell line is required along with plasmid DNA and transfection agents that increase COGS for each batch. Stoichiometry and expression become important when there are multiple plasmids used (helper and transfer).
Spodoptera frugiperda (Sf9) insect cells using the baculovirus expression vector system (BEVS) for transfection. During a manufacturing run, baculoviruses (one for rep/cap and the other for the AAV genome) with the transfer genes to generate the AAV vectors are infused with the Sf9 cell culture. Each baculovirus requires a master line, which increases the upfront investment required versus HEK292. However, yields in an Sf9 system are significantly higher.
A major theme in gene therapies is that these processes are the actual product. Ultimately, the goal is to develop cell-lines that maximize viral titers (i.e. produce fewer empty capsids) and minimize upfront development costs (a batch can cost upwards of $500K). However, the AAV manufacturing field has 3 major problems to surmount:
Developing stable cell lines to increase yields - the current standard for vector production relies on transient transfection, mainly to reduce costs
Minimize the variability between batches, serotypes, and cargos - depending on the serotype and what is being delivered can lead to wide variations in outcomes across manufacturing runs
Reduce the number of empty capsids produced - cell lines produce many non-functional empty capsids that need to be separated from functional capsids
Improve purification - recovering AAV vectors from culture supernatant versus cell lysate. Moreover, generic processes particularly around purification are needed to apply across multiple vectors.
Over time, there is a large opportunity to design and scale vector-specific manufacturing processes. With more than 12 AAV serotypes, 100s of natural variants, and many more synthetic versions, the same focus and specificity required for drug development is needed in biomanufacturing.
Source: Piper Jaffray
iPSC-derived cell therapies
Induced pluripotent stem cells (iPSC) have the ability to differentiate into any cell type and have the power to self-renew. These features make them particularly compelling for regenerative medicine and allogeneic cell therapies.
The key work was led by Shinya Yamanaka at Kyoto University to discover and use 4 reprogramming factors (OCT4, SOX2, MYC, and KLF4) to induce pluripotency from mouse embryonic or adult fibroblasts - https://www.cell.com/cell/fulltext/S0092-8674(06)00976-7 Before this work was published in 2006, the stem cell field relied on human embryonic stem cells (ESC), which are difficult to source and historically has controversy surrounding the research. Once Yamanaka published this research, it became clear that iPSCs can solve these sourcing and ethics problems that have slowed down progress in the field. Rather than sourcing an ESC for each barch, one can create a iPSC master cell line.
Before 2006, some early success of ESCs in clinical trials - (1) https://pubmed.ncbi.nlm.nih.gov/29553577/ and (2) https://pubmed.ncbi.nlm.nih.gov/27144379/ have set up iPSC-derived cell therapies to have some sort of precedent and hopefully get translated into new medicines sooner rather than later - https://ir.fatetherapeutics.com/news-releases/news-release-details/fate-therapeutics-announces-fda-clearance-ind-application-ft596
Like most new inventions, iPSCs face a host of new challenges to solve. Currently, iPSCs have a longer road to travel and a few key problems to solve:
Teratoma formation - pluripotent stem cells can form teratomas in a patient due to the cancer risk of the 4 Yamanaka factors. A useful strategy is to purge residual iPSCs before a transplant.
Carcinogenicity - related to (1), expression of the 4 factors, in particular MYC and KLF4, increase the odds of tumor formation. Finding substitute genes that reduce this risk but still induce pluripotency is needed: Nanog and LIN28 might be able to substitute for MYC and KLF4.
Genomic instability - there is evidence that reprogramming a cell itself can lead to generic instability (i.e. amplifications, deletions) as well as chromosome and epigenetic abnormalities. This is still an important area of research with a focus on the role of p53 in reprogramming.
Immunogenicity - iPSCs can lead to immune rejection depending on the tissue and minor antigen presented. One strategy is to use genome engineering to knockout diverse HLA genes and activate immunomodulatory genes to develop hypoimmunogenic iPSCs (i.e. the basis of Sana) - https://www.pnas.org/content/116/21/10441.abstract
Quality control - manufacturing iPSCs consistently and reproducibly is needed. Better tools to isolate, select, and expand certain iPSC lines are needed.
Increase the efficiency of differentiation - new factors and differentiation protocols are needed to reduce the number of residual iPSCs within a batch and hopefully reduce the time from months to weeks
Ultimately, iPSC-derived cell therapies have the potential to create master cell lines that can be used off-the-shelf to treat a large number of patients from oncology to regeneration.
Source: Fate Therapeutics
Voice in healthcare
Everything in healthcare from diagnostics to prescriptions starts with a conversation between a patient and their doctor. Given the massive improvements in voice recognition and transcription technology, with the best example in Alexa, bringing these tools to healthcare can help patients remember the conversations they have with their doctor, help physicians save time, and reduce billing errors.
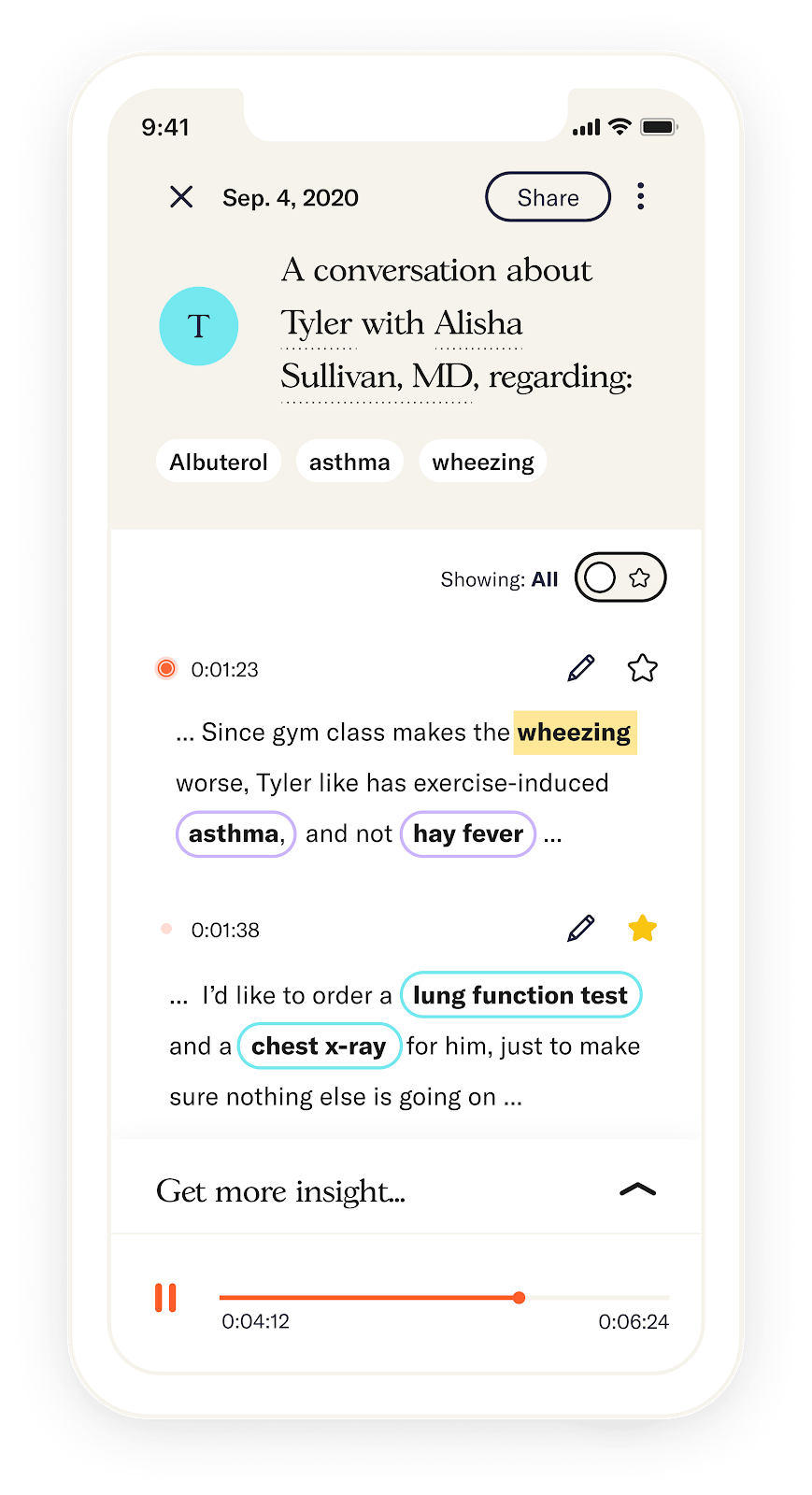
The two leading companies in the field are Abridge and Suki. Both are building similar products to track and translate the conversations between a patient and doctor into actionable results, but Abridge is focused on acquiring consumers and Suki on healthcare providers.
For the patient:
A voice recording product can help them control part of their own health records. They could share their conversations with doctors with other clinicians and care providers.
They more easily remember details from their conversations. Like what medications they need to take and why. Their diagnoses and more.
Increasing compliance rates (i.e. filling their prescriptions, follow-ups)
Source: Abridge
For the doctor:
Reduce the time they spending taking medical notes and increase their time with patients by transcribing a conversation into an EHR
Help retrieve certain medical information like charts more easily
Condense a conversation they have with a patient into an actionable plan
Integrate with other service providers like PBMs to automatically prescribe a medicine to their patient
Increase the quality of treatment plans and information on their patient, to reduce claim denials
Building voice products in healthcare has the potential to introduce virtual assistants to the exam room. In combination with machine learning, these products can become personalized to both the patient and doctor. Technical challenges remain to define the large universe of medical terms and implement algorithms that can recognize patient-relevant words while accommodating for differing qualities of microphones in the exam room. Privacy and security is also incredibly important.
N-of-1 trials
N-of-1 trials focus on understanding the effects of various interventions for an individual patient. In 1986, a group out of McMaster University published the first paper defining N-of-1 trials inspired by a drug trial for asthma - https://www.nejm.org/doi/pdf/10.1056/NEJM198604033141406 In this type of trial, a patient alternates between taking a drug and placebo. These treatments can be sequenced in various ways to establish the effects of the drug. After the trial, statistical analysis tools can be used to determine the efficacy of the drug and the one trial can be aggregated with other N-of-1 trials to infer the drug’s efficacy across a population.
N-of-1 trials are valuable for their ability to capture intra-individual variation. Other trial designs are good for inter-individual diversity and meet their limitations where patient responses to an intervention have high variation. N-of-1 become useful in situations where a randomized clinical trial (RCT) cannot address:
Diseases with low prevalence (i.e. rare diseases)
Diseases with large response variability (i.e. chronic pain)
Patients who have been on long-term treatment regimens
Patients who are medical outliers
In short, N-of-1 trials can make drug development for rare diseases feasible where thousands have no approved medicine. Moreover, N-of-1 trials can act as an early signal on where to engage in a costly RCT and identify predictors of response beforehand. Ensuring trial quality across patients and comprehensive reporting can help N-of-1 trials to gain more acceptance from the medical community. Two large opportunities are to bring the power of antisense oligonucleotides (ASO) to N-of-1 trials (invest upfront and costs go down overtime) and build better software/data tools to capture data from these trials and put them into an EHR so results can be aggregated and analyzed with confidence by clinicians.
Patient-driven drug development
Early-stage drug development, particularly in rare diseases, have high risks of failure or are too small of markets to attract substantial venture capital. As a result, foundations and patients have been groups that fill in this gap. With better tools to build communities and precedents set, patients can have an increasingly powerful role in starting new companies and sparking the development of medicines for their disease. Opportunities in the field are:
Building a network of patients and their data - using a Flatiron-type of model to collect natural history of patients, especially those with rare diseases.
Aggregating foundations - to provide resources like patient support, help with trial recruitment, and understanding of new and emerging treatments
Hand off to industry - foundations and patient groups are experts in their disease and help industry understand how the field is developing. The Parker Institute for Cancer Immunotherapy does a great job here.
Minimizing conflicts-of-interest - foundations and companies need to find a balance to manage actual and perceived conflicts-of-interest especially if foundations take equity stakes in the companies
Monetizing equity and royalties - finding efficient pathways for foundations and patient groups to profit from their early investment in order to reinvest the capital into their non-profit activities. The Cystic Fibrosis Foundation and Aurora Biosciences, which was acquired by Vertex and led to the approval of Kalydeco in cystic fibrosis is the best example of this.
Translating research into products - many diseases, rare diseases mainly, do not have an established scientific understanding. As a result, foundations have to fill in the gap and invest in basic research to set up drug development for these diseases down the line.
The best example so far of patient-driven drug development is the Cystic Fibrosis Foundation (CFF) and Kalydeco. CFF’s mission is to cure cystic fibrosis (CF), a rare disease that affects ~40K people in the US. The CFF was formed in 1955 and spent decades funding basic research in CF and building a database of patients. In 2000, the foundation invested $30M in Aurora Biosciences to develop new medicines for CF targeting a mutation discovered by labs in the 1980s funded by the CFF. Vertex acquired Aurora in 2001 and the CFF maintained their drug development agreement leading to the approval for Kalydeco in 2012. In 1980, the median age of survival for a CF patient was 18 and by 2016 that age of survival increased to well above 45 years old. Inspiring work by the CFF and serves as a case study for the power of patient-driven drug development. The foundation continues to support the development of new medicines with Orkambi (a combination of Kalydeco and Lumacaftor) recently approved for CF and ensure patient access to these transformative drugs.
Other examples of successful patient-driven drug development are LifeArc and Keytruda along with the Juvenile Diabetes Research Foundation, Emerson Collective, PICI, and the Michael J. Fox Foundation. These groups have the power to lower the barriers for drug development in particular diseases by providing patients, scientific expertise, and capital.