Axial partners with great founders and inventors. We invest in early-stage life sciences companies such as Appia Bio, Seranova Bio, Delix Therapeutics, Simcha Therapeutics, among others often when they are no more than an idea. We are fanatical about helping the rare inventor who is compelled to build their own enduring business. If you or someone you know has a great idea or company in life sciences, Axial would be excited to get to know you and possibly invest in your vision and company . We are excited to be in business with you - email us at info@axialvc.com
Inventors #9
A set of ideas and observations on inventions and discoveries in life sciences.
Immunology
The immune system and everything in it.
Gβγ is a direct regulator of endogenous p101/p110γ and p84/p110γ PI3Kγ complexes in mouse neutrophils - https://stke.sciencemag.org/content/13/656/eaaz4003 - the Hawkins Lab at Babraham Institute determined the role of PI3Kγ (a target important for myeloid leukemias) subunits in activating neutrophils (make up the majority of white blood cells):
The group created mouse models with mutations that prevented Gβγ (a GPCR subunit) from binding either one of PI3Kγ’s 2 subunits: p110γ (catalytic) and p101 (regulatory)
It’s not clear to me why they chose to study the role of these interactions in neutrophils (I think because PI3Kγ plays a significant role in macrophage and neutrophil activation); however, the group observed lower migration rates and activation levels after agonist treatment. This effect is more pronounced for p110γ mutants.
The p110γ subunit alone was insufficient to activate GPCRs from the Ras pathway. This is a pretty important discovery since it's in vivo (a first) and gets to an MoA for PI3Kγ-driven activation of neutrophils.
This is an important step to understand how GPCRs within the same pathway can generate several responses and opens up new avenues to drug PI3Kγ in oncology
Biochemistry and structural biology
The granddaddy of them all.
Complex Crystal Structures of EGFR with Third-Generation Kinase Inhibitors and Simultaneously Bound Allosteric Ligands - https://pubs.acs.org/doi/10.1021/acsmedchemlett.0c00472 - the Rauh Lab from TU Dortmund determined the structure of EGFR in complex with the standard-of-care (SoC) for NSCLC and an allosteric inhibitor to understand the structural mechanism to overcome resistance:
In NSCLC, the SoC is Osimertinib; however the C797S resistance mutation (found in ~10% of patients) that converts from a cysteine to a serine to make the covalent anchor point less reactive
To overcome this resistant mutation, allosteric inhibitors can be used to bind different sites of EGFR
The group used X-ray crystallography to determine the structures of EGFR in complex with different inhibitors binding the active site in combination with allosteric inhibitors
These provide the foundation to use structure-based design for new EGFR inhibitors to overcome the C797S mutation
Neuroscience
Roughly 20 years behind but set up to transform the concept of human.
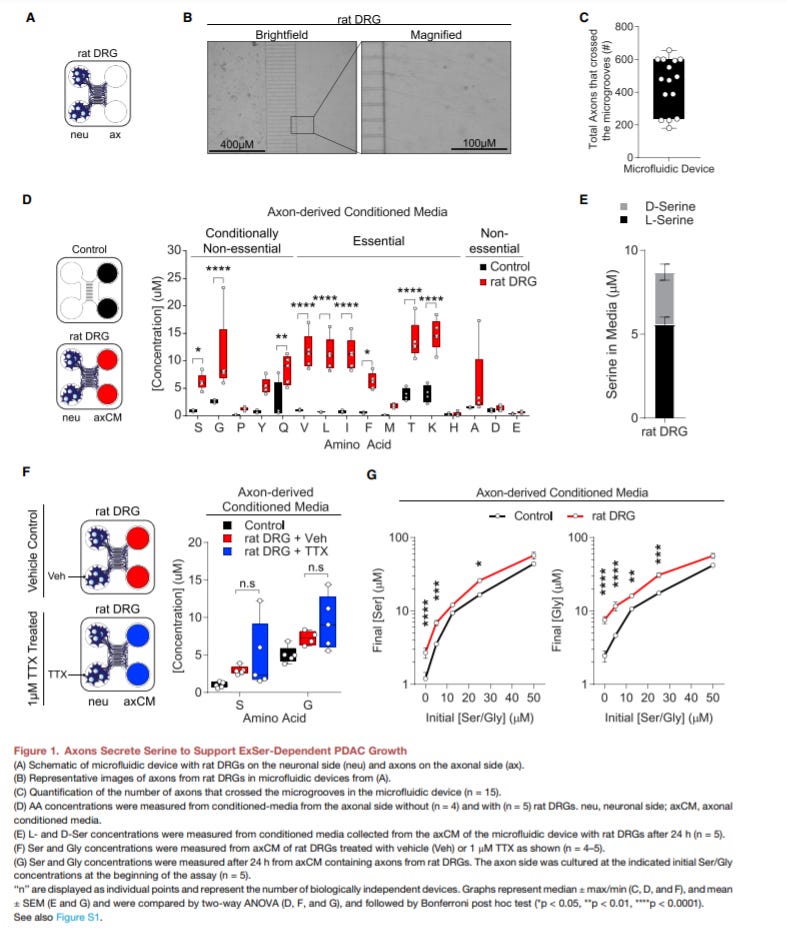
Neurons Release Serine to Support mRNA Translation in Pancreatic Cancer - https://www.cell.com/cell/fulltext/S0092-8674(20)31322-2 - the Kimmelman Lab from NYU Langone put out work connecting the nervous system and pancreatic cancer where nerves surrounding the tumor secrete serine to promote tumor growth:
Pancreatic ductal adenocarcinomas (PDAC) are highly innervated. The group had the hypothesis that PDACs have higher metabolism rates despite a nutrient-poor microenvironment, nerves might be a source of nutrients for the cancer cells. Moreover, a large portion of PDAC cells lack certain serine biosynthesis pathway proteins creating nutrient-poor regions within a tumor.
The group discovered that under serine deprivation conditions, PDAC cells secrete nerve growth factor (NGF) to stimulate surrounding nerves to release serine
Blocking tumor innervation with a Trk-NGH inhibitor decreased PDAC growth
This paper established the role of the peripheral nervous system in pancreatic cancer as well as connecting it to a mechanism: serine availability
Cell biology
Cell structure and function.
MLL4-associated condensates counterbalance Polycomb-mediated nuclear mechanical stress in Kabuki syndrome - https://www.nature.com/articles/s41588-020-00724-8 - from the Zippo Lab at the University of Trento, the group determined that haploinsufficiency of MLL4 (lysine methyltransferase) contributes to Kabuki syndrome (a rare disease) through significant reorganization of transcriptional condensates and the physical properties of the nucleus:
The group studied how the loss-of-function of MLL4 changes chromatin compartmentalization through liquid-liquid phase separation
Using a Kabuki syndrome mesenchymal stem cell model, the paper found that MLL4 is a significant driver for the proportion of transcription-associated and polycomb (repressive) condensates. Blocking ATR, a mechanosensor in the nucleus, rescues the effects of losing MLL4 function leading to normal stem cell differentiation.
This is a pretty important finding to connect nuclear condensates to stem cell differentiation. The group has genetic data to allude that mechanical stress within the nucleus is a key driver but this is a further area of study.
In Kabuki syndrome, MLL4 levels are cut in half leading to dysfunctional condensate formation in the nuclear. This might be a new MoA for the disease. Inhibiting ATR might be a therapeutic avenue to pursue as well as general strategies to develop new medicines that open chromatin and increase transcription-associated condensates.
Genetics, genomics, and developmental biology
Heredity and variation.
Arming Yourself for The In Silico Protein Design Revolution - https://www.cell.com/trends/biotechnology/fulltext/S0167-7799(20)30267-5 - a review from the Tangney Lab at University College Cork does an incredible job laying out the opportunity for de novo design to transform the study of proteins and their development into new products:
There are around 10^12 natural proteins with 20^x possible variants where x is the number of aminos acids in a protein
There are two approaches in the field: (1) repurposing existing protein scaffolds (2) creating synthetic proteins with new functions
Recent examples are LOCKR - https://www.ipd.uw.edu/2019/07/introducing-lockr-a-bioactive-protein-switch/
AlphaFold from DeepMind - https://deepmind.com/blog/article/AlphaFold-Using-AI-for-scientific-discovery
UniRep for protein structure prediction - https://www.nature.com/articles/s41592-019-0598-1
With so much search space, the key to success in de novo protein design is reducing search space that can be manageably screened