Axial - Inventors #5
Surveying great inventors and businesses
Axial partners with great founders and inventors. We invest in early-stage life sciences companies such as Appia Bio, Seranova Bio, Delix Therapeutics, Simcha Therapeutics, among others often when they are no more than an idea. We are fanatical about helping the rare inventor who is compelled to build their own enduring business. If you or someone you know has a great idea or company in life sciences, Axial would be excited to get to know you and possibly invest in your vision and company . We are excited to be in business with you - email us at info@axialvc.com
Inventors #5
A set of ideas and observations on inventions and discoveries in life sciences.
Immunology
The immune system and everything in it.
Illuminating Epigenetics and Inheritance in the Immune System with Bioluminescence - https://www.cell.com/trends/immunology/fulltext/S1471-4906(20)30208-8 - the Fisher Lab from the London Institute of Medical Sciences, did a really good job to lay out the potential for the use of bioluminescence imaging (i.e. mainly different luciferase/luciferin combinations) in immunology:
Opportunities to develop new bioluminescent systems - discover new luciferases, new substrates, developing multiple reporter systems to image multiple targets/pathways simultaneously, and beyond - https://basicmedicalkey.com/engineering-luciferases-for-assays-and-imaging/
Using gene editing tools to design and implement reporter constructs more quickly
CRISPR in particular enables easier insertion of reporter constructs into endogenous loci; helps generating transgenic mice much easier
Enabling noninvasive imaging of changes in immune cell gene expression in vivo. In many situations, immune cells have short half lifes (i.e. neutrophils) and spatial/temporal changes in activity. The paper argues that bioluminescence imaging can more easily track immune cells across their development and lifespan. An example they use, is the power of a dual reporter system to measure both T-cell localization and activation to understand tissue/site selectivity.
Biochemistry and structural biology
The granddaddy of them all.
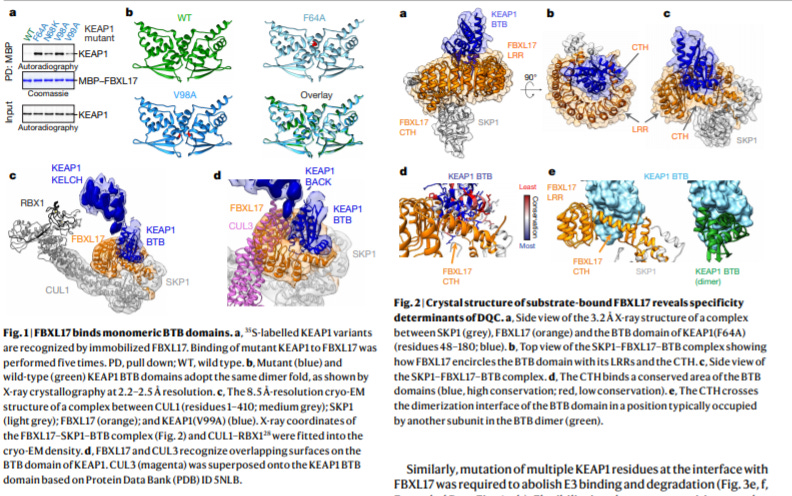
Structural basis for dimerization quality control - https://www.nature.com/articles/s41586-020-2636-7 - from the Rape Lab at UC Berkeley along with Eva Nogales for her cryo-EM expertise and John Kuriyan for biochemisty, the paper does a great job at understand how the SCF–FBXL17 E3 ligase selectively probes for heterodimers for proteins with the BTB domain. SCF–FBXL17 selectively degrades heterodimers of BTB proteins. The first key experiment was generating mutants of a SCF substrate and demonstrating the ligase’s ability to not ubiquitinate wildtype and the substates lack of BTB structural changes suggesting SCF probes via another mechanism. The second key experiment was to use cryo-EM to solve the structure discovering that the E3 ligase doesn’t bind the heterodimer binding site but a distinct site on the singel BTB domain. The authors suggest that SCF measures BTB dissociation rates within a heterodimer.
Neuroscience
Roughly 20 years behind but set up to transform the concept of human.
Multiscale and multimodal reconstruction of cortical structure and function - https://www.biorxiv.org/content/10.1101/2020.10.14.338681v1 - out of the Seung Lab at Princeton, the preprint reports the reconstruction from electron microscopy of parts of the mouse visual cortex coupled with 2-photon calcium imaging of pyramidal cells. What makes this resource paper particularly exciting is that past studies were limited to 10s of cells while this paper records 100s from pyramidal cells to astrocytes and microglia as well as ~180K mitochondria within some of these cells, which seems to be enabled by cheaper computation and better electron microscopes:
Data is at microns-explorer.org
An interesting discovery is that mitochondria coverage within cell and synapse density is correlated and might be location-specific
Cells with more incoming connections have more reliable visual responses
Overall, this paper shows the potential power of connectomics to discover new classes of biology in neuroscience
Cell biology
Cell structure and function.
Organelle membrane-specific chemical labeling and dynamic imaging in living cells - https://www.nature.com/articles/s41589-020-00651-z - the Hamachi Lab at Kyoto University developed a useful chemical probe to selectively label and image lipids (phosphatidylcholine) in specific organelles. They image lipids in the mitochondria and ER/Golgi intermediate compartment; the key breakthrough is that even though lipids can be chemical labeled, tracking them for a specific cell compartment is not possible for “real-time” imaging. The authors simply split up the probe with one half for the lipid and the other half a organelle-specific probe (fluorescent) that binds with the lipid label via click chemistry. With this premise, the authors do some exciting work to image lipids with the mitochondria and ER/Golgi. Chemical probes open up new avenues for biology. So the expansion of more of these probes ought to uncover some interesting observations.
Genetics, genomics, and developmental biology
Heredity and variation.
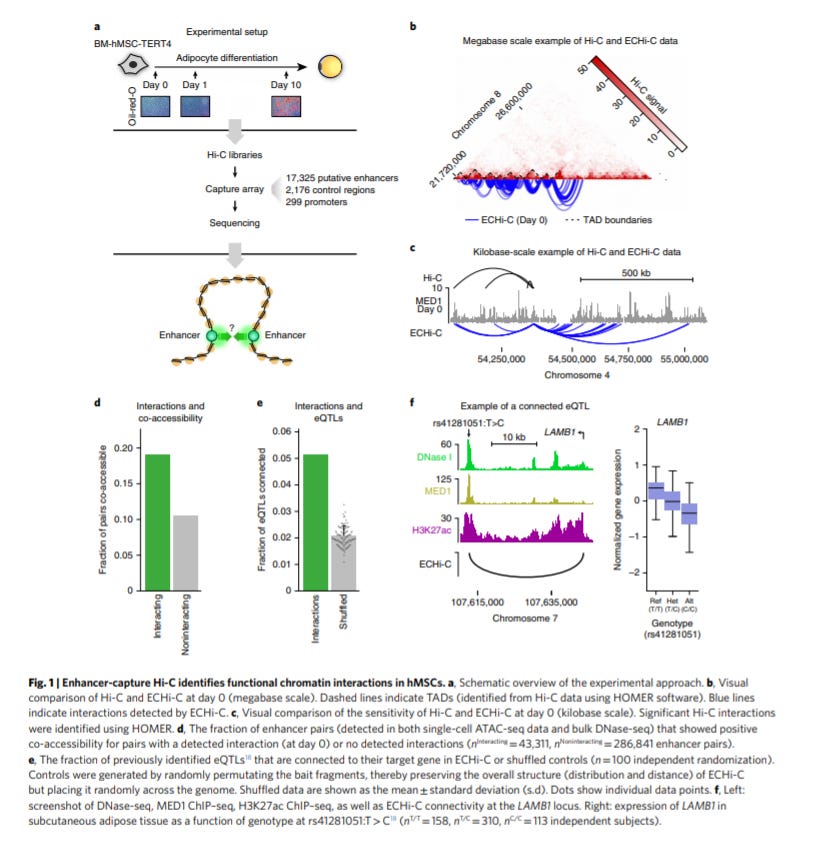
Highly interconnected enhancer communities control lineage-determining genes in human mesenchymal stem cells - https://www.nature.com/articles/s41588-020-0709-z - the Mandrup Lab out of the University of South Denmark map out enhancer/enhancer interactions during human mesenchymal stem cells. The paper is a great example of the role of the 3D genome in core biological phenomena and the power of genomics to identify key components of the 3D genome in a disease or pathway. The premise is that highly interconnected enhancers (HICE) act as groups to promote engagement of target enhancers and thereby their genes and consequently, decrease the activity in other HICEs. The key tool used is Hi-C to measure the 3D genome but the group focuses on mapping out promoters/enhancers. The group chooses to study adipocyte differentiation, across 4 time points, (maybe because of a grant) to understand how HICEs drive cell lineages:
During adipocyte differentiation, certain HICE groups are either activated or repressed
Phase-separated domains form around genes that determine cell lineage
HICEs coverage on these lineage-determining genes and their promoters
Next steps are to understand how transcription factors activate certain HICEs and the mechanism by which HICEs are repressed