Axial partners with great founders and inventors. We invest in early-stage life sciences companies such as Appia Bio, Seranova Bio, Delix Therapeutics, Simcha Therapeutics, among others often when they are no more than an idea. We are fanatical about helping the rare inventor who is compelled to build their own enduring business. If you or someone you know has a great idea or company in life sciences, Axial would be excited to get to know you and possibly invest in your vision and company . We are excited to be in business with you - email us at info@axialvc.com
Inventors #18
A set of ideas and observations on inventions and discoveries in life sciences.
Immunology
The immune system and everything in it.
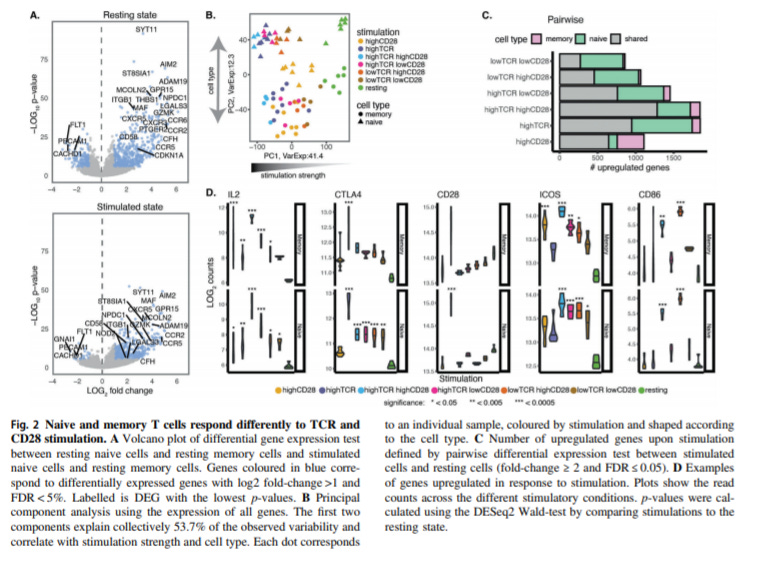
Genomic profiling of T-cell activation suggests increased sensitivity of memory T cells to CD28 costimulation - https://www.nature.com/articles/s41435-020-00118-0 - the Trynka Lab at the Wellcome Sanger Institute discovered that memory T-cells are more sensitive to CD28 costimulation versus naive T-cells:
T-cells require 2 signals to become activated: one from a T-cell receptor (TCR) and the other from a co-stimulatory signal
The most well characterized co-stimulatory signal is CD28, which interacts with B7 expressed on antigen-presenting cells (APC). CD28 is mainly important to activate T helper cells (CD4+)
The paper profiled gene expression and chromatin accessibility of T-cells with varying TCR and CD28 signalling to understand the impact on co-stimulation of particular subsets of CD4+ T-cells
A key part of their experimental design was only profiling stimulated T-cells (using flow) then separately assessing whether they were naive (CD45RA+) or memory T-cells (CD45RA-)
First, the group discovered that memory T-cells are more sensitive to CD28 costimulation while naive T-cell stimulate is more driven by TCR activation
Profiling chromatin accessibility revealed that the AP-1 transcription factor is more likely to be enriched on genomic sites when both a TCR and CD28 activate a T-cell
The paper also discovered a series of genes sensitive to CD28 engagement, almost all non-coding, that could influence the expression of targets like STAT1 and IL7 and an overall immune response
Costimulation is incredibly important for an immune response and understanding what signals are required and at what intensities can inform new therapeutic development in cancer immunotherapy and autoimmunity
Biochemistry and structural biology
The granddaddy of them all.
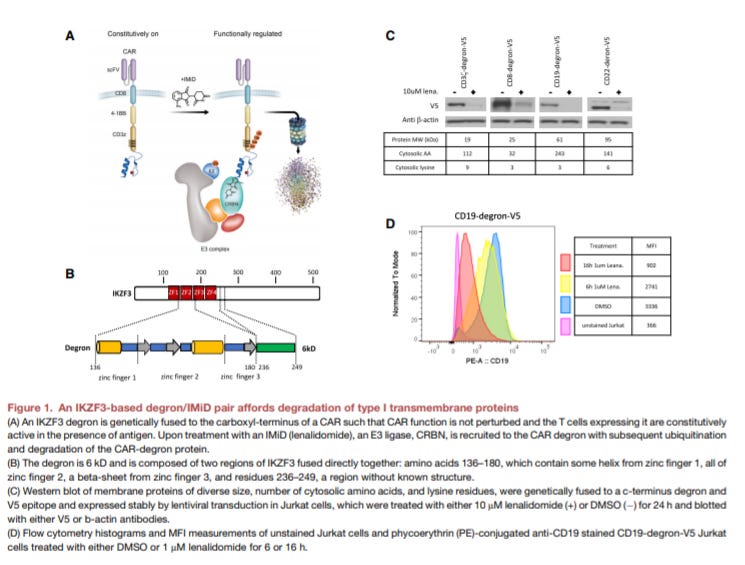
An IMiD-inducible degron provides reversible regulation for chimeric antigen receptor expression and activity - https://www.cell.com/cell-chemical-biology/fulltext/S2451-9456(20)30475-X - a group at Novartis led by Carla Guimaraes invented a ligand controlled chimeric antigen receptor (CAR):
CAR-T cell therapies have been transformative for patients with certain blood cancers and have the potential to bring curative treatments to oncology and autoimmunity
Despite the promising results, these medicines sometimes have severe side effects like neurotoxicity and CRS. So the motivation for the work here was to find a way to control CAR-T activity with an FDA approved drug
So the group designed a CAR with a understand the control of a IKZF3 ZF2 b-hairpin IMiD-inducible degron, which had been previously used to control the expression of other membrane proteins and is under the control by lenalidomide, an FDA-approved chemotherapy
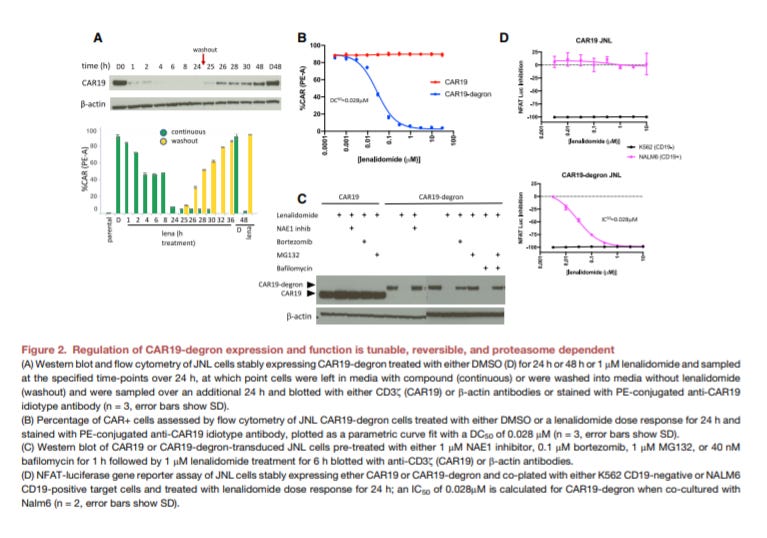
Using a CAR-T targeting CD19 (CAR19), the group found that upon treatment of lenalidomide, the CAR experienced rapid degradation with ~80% of the population gone within an hour and restoration to normal expression levels after ~10 hours. Using flow cytometric, CAR19 cells with the degron system were found to only respond to the drug treatment. They also found that these cells had around 50% lower surface CAR19 molecules than normal CAR-T cells probably due to uncharacterized interactions between the degron and other ubiquitin ligases.
To test the efficacy of the new CAR19 system, mouse models for ALL were used: the CAR19 cells with the degron were given to mice 7 days after a tumor transplant and given lenalidomide 5 days after. They found that tumor growth increased versus normal CAR-T treatment a few days after this treatment.
This proof-of-concept study showed the ability to tune CAR activity in vivo; however, understanding how dosing changes for this type of drug candidate as well as studying the comparable functionality of a ligand-controlled CAR are the next steps
Neuroscience
Roughly 20 years behind but set up to transform the concept of human.
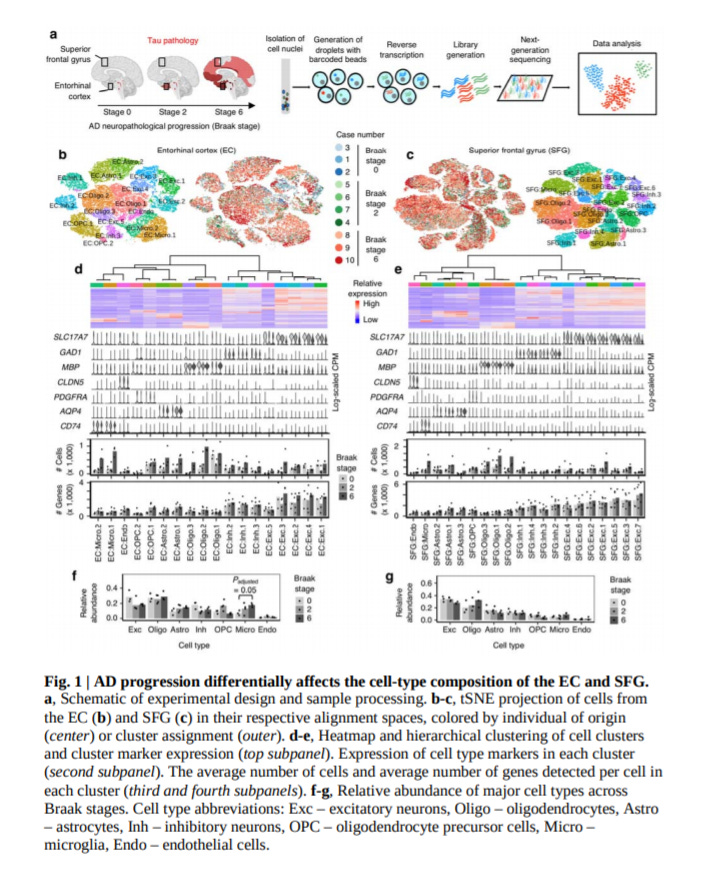
Molecular characterization of selectively vulnerable neurons in Alzheimer’s Disease - https://www.nature.com/articles/s41593-020-00764-7 - the Kampmann Lab at UCSF profiled postmortem brain tissue from 10 patients spanning the range of Alzheimer’s Disease (AD) neuropathology RNA-seq to discover new cell populations driving the disease:
This work identified: (1) a biomarker for susceptibility to neuronal dysfunction, RORB, specific to entorhinal cortex excitatory neurons and (2) another biomarker, GFAP, for an astrocyte subpopulation
RORB is a receptor found in parts of the human cortex; neurons with this market had a ~50% decline in abundance at the early stages of AD. Tau neurofibrillary inclusions were observed to have accumulated in these specific neurons as well. Further studies using microscopy to couple morphological changes with sequencing are needed to find potential classes of vulnerable inhibitory neurons.
The authors of the study reported that these sequencing efforts had low coverage for microglia; however, an astrocyte population expressing GFAP in the cortex were found to be vulnerable to dysfunction in AD.
A limitation of this study was focused on males with a specific genetic background - APOE ε3/ε3. Expanding to more diverse patients
Next steps are to build models (i.e. animal, iPSC) to discover the mechanisms by which GFAP and RORb act to imbue selective vulnerability. This work could set the basis for new medicines to treat AD and neurodegeneration in general.
Cell biology
Cell structure and function.
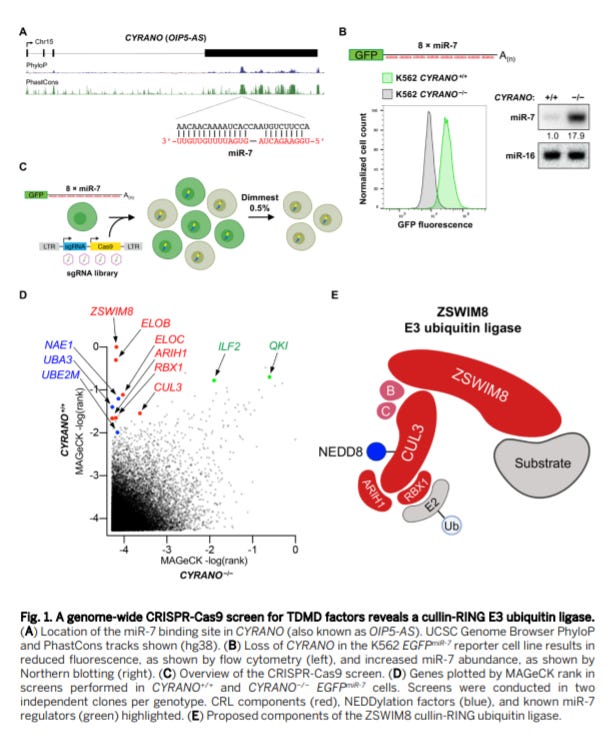
A ubiquitin ligase mediates target-directed microRNA decay independently of tailing and trimming - https://science.sciencemag.org/content/370/6523/eabc9546 - out of the Mendell Lab at UT Southwestern, the group discovered an ubiquitin ligase that mediates microRNA (miRNA) degradation:
miRNAs have the ability to inhibit multiple (somewhat) complementary RNA targets through inhibition of translation or target degradation
If a miRNA encounters a highly complementary target, target-directed microRNA degradation (TDMD) occurs
The goal of the study was to determine the mechanism by which miRNAs are directed to degradation during TDMD
The group discovered a cullin-RING ubiquitin ligase (CRL) that mediates miRNA degradation. A genome-wide CRISPR screen was used in K562 cells to discover genes that decrease the expression of miR-7 (measured by enhanced GFP).
This unbiased screen discovered the CRL that when knocked out led to a significant decrease in miR-7 levels. Various studies characterized the ligase’s interaction and effects on other miRNAs. Next steps are to identify if the ligase mediates TDMD through ubiquitylation of the microRNA’s cognate Argonaute protein or by other means? Does the CRL recruit other proteins to destabilize the miRNA?
Genetics, genomics, and developmental biology
Heredity and variation.
The pyruvate-lactate axis modulates cardiac hypertrophy and heart failure - https://www.sciencedirect.com/science/article/pii/S1550413120306586?dgcid=coauthor - the Drakos Lab at the University of Utah use genetics to connect the pyruvate-lactate axis (PLA) to cardiac hypertrophy:
Like any good Cell paper, the work conducted a series of well-thought out experiments in humans and mouse models to investigate the role of pyruvate/lactate (important molecules in metabolism) in heart failure (HF)
First, patients with chronic HF have lower levels of mitochondrial pyruvate carrier (MPC) in heart tissue and HF patients who recovered after a left ventricular assist device (LVAD) implantation had an increase in MPC levels. This observation led the authors to hypothesize that changes in metabolism could influence the recovery of a HF patient. This effect by MPC was confirmed in models for cardiac recovery.
MPC deficiency in heart tissue in mice led to cardiac hypertrophy and HF. This effect was also observed in cardiomyocytes.
The counterbalance to MPC during metabolism is monocarboxylate transporter 4 (MCT4). With a MCT4 inhibitor, the group restored pyruvate flux in the mitochondria and attenuated hypertrophy is cardiomyocytes
This work establishes the pyruvate-lactate axis, in particular MCT4, as a new target to treat HF and help with myocardial recovery