Axial partners with great founders and inventors. We invest in early-stage life sciences companies such as Appia Bio, Seranova Bio, Delix Therapeutics, Simcha Therapeutics, among others often when they are no more than an idea. We are fanatical about helping the rare inventor who is compelled to build their own enduring business. If you or someone you know has a great idea or company in life sciences, Axial would be excited to get to know you and possibly invest in your vision and company . We are excited to be in business with you - email us at info@axialvc.com
Inventors #13
A set of ideas and observations on inventions and discoveries in life sciences.
Immunology
The immune system and everything in it.
iPSC-derived NK cells maintain high cytotoxicity and enhance in vivo tumor control in concert with T cells and anti–PD-1 therapy - https://stm.sciencemag.org/content/12/568/eaaz5618 - the Miller Lab at the University of Minnesota along with help from Fate Therapeutics showed that iPSC-derived NK cell synergize with anti-PD-1 antibodies and the adaptive immune system in vivo to reduce tumor burden and increases cytotoxicity in cells lines:
Versus T-cells, NK cells are ideal for allogeneic (off-the-shelf) cell therapies because they do not required HLA matching for target cell killing
However, manufacturing therapeutically-sufficient NK cells has been a barrier for use; the use of induced pluripotent stem cells (iPSC) offers a pathway to generate a renewable source of NK cells
The paper set out to understand the role of iPSC-derived NK cells (iNK) in checkpoint inhibition and the former’s potential to increase response rates
The group showed that after successful isolation and expansion of CD3-CD56+ NK cells, that they were phenotypically and transcriptionally similar to peripheral blood - secreting cytokines and generating cytotoxicity in various cancer cell lines
In a xenograft model for ovarian cancer, the iNK cells were shown to extend survival rates, and in tumor spheroids, the cells synergized with an anti-PD-1 antibody to produce cytokines and reduce spheroid volume
The idea is that the iNK cells mediate these effects not only through direct cytotoxicity but T-cell recruitment and activation in the microenvironment. So iNK cells act as a link between the innate and adaptive immune systems - subverting resistance to checkpoint inhibitors and increase patient responses
The potential for using NK cells is promising but manufacturing and sourcing material is where advantages can be obtained: iPSCs can be used along with cord blood and engineered cell lines. This group has initiated a phase 1 trial using their iNK cells to treat lymphoma and solid tumor patients who had not responded to checkpoint inhibition - https://clinicaltrials.gov/ct2/show/NCT03841110
Biochemistry and structural biology
The granddaddy of them all.
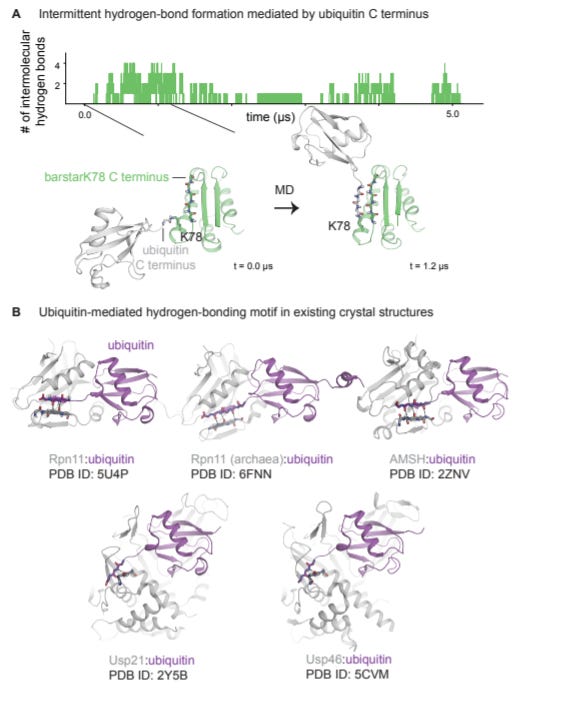
Mechanistic basis for ubiquitin modulation of a protein energy landscape - https://www.biorxiv.org/content/10.1101/2020.12.06.414011v1 - from the Marqusee Lab at UC Berkeley, the group studied the destabilizing (and stabilizing) effects of ubiquitination on target proteins:
Ubiquitin is a post-translational modification (PTM) that is mainly used to guide proteins to degradation through the proteasome
The group sought to understand the biophysical effects of ubiquitination on the target protein and how its effects depend on the site
Previously the group used barstar as a model protein to find that ubiquitination (Ub) at K2 and K60 destabilized the protein while Ub at K78 had little-to-no effect on the protein’s stability
For this paper, the same model and targets were used in combination with NMR, mass spec, and molecular dynamics to find that ubiquitin at destabilizing sites causes more exposure of the target protein’s C-terminus rather than some large-scale structure rearrangement. This mechanism involves a combination of ubiquitin reducing the target protein’s entropy (K60) or of enthalpy (K2). Whereas Ub at K78 actually formed a structure to protein the C-terminus.
This work is an important step to understand the physical impact of site-specific ubiquitination and its role for targeted protein degradation
Neuroscience
Roughly 20 years behind but set up to transform the concept of human.
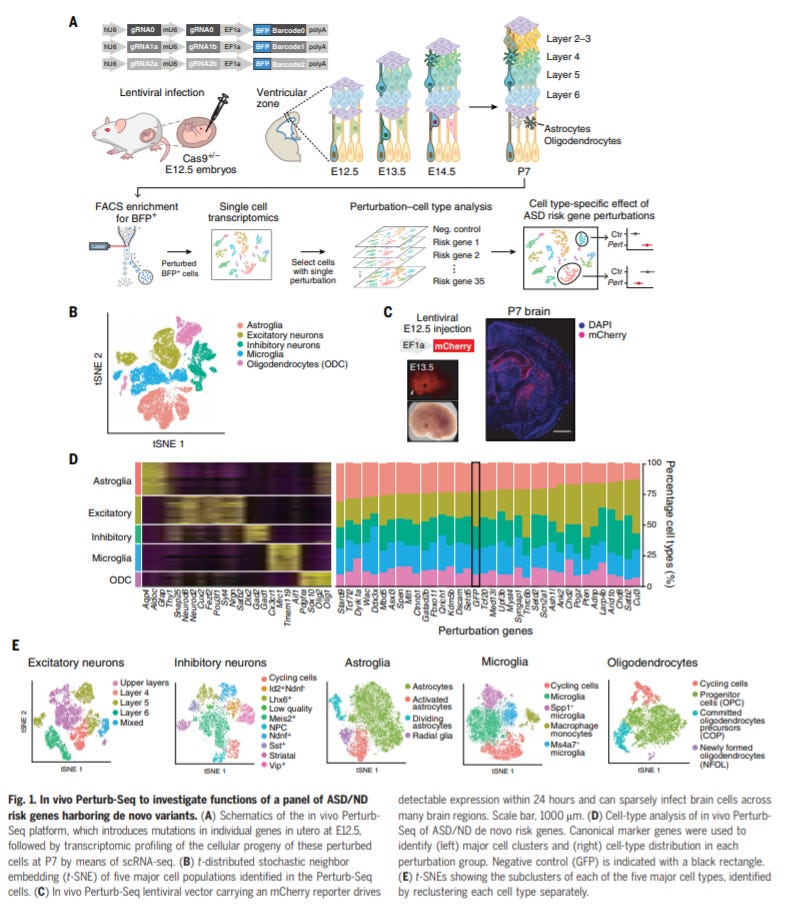
In vivo Perturb-Seq reveals neuronal and glial abnormalities associated with autism risk genes - https://science.sciencemag.org/content/370/6520/eaaz6063 - the Arlotta Lab at Harvard used Perturb-seq to screen 35 autism spectrum disorder (ASD) genes in vivo:
Many studies have connect certain genetic variants to neurological diseases; however, comprehensively screening across these genes has been a challenge
As a result, the large used a new invention (~4 years old), in vivoPerturb-seq to study 35 ASD genes in mice: using CRISPR gene editing to generate genetic mutations in the neocortex of embryonic mice and RNA-seq’ed the brain of the birthed mice
This work unveiled new genetic modules and validated existing ones for ASDs - (1) validating Ank2’s , an ankyrin protein involved in membrane scaffolding, role in excitatory neurons’ function as well as finding new roles in interneurons and astrocytes (2) Chd8, a Wnt signalling modulation, is significant for oligodendrocyte maturation
Cell biology
Cell structure and function.
Spatial Multiplexing of Fluorescent Reporters for Imaging Signaling Network Dynamics - https://www.cell.com/cell/fulltext/S0092-8674(20)31399-4 - out of the Boyden Lab at MIT, the group invented a new reporter tool, signalling reporter islands (SiRI), to measure multiple proteins and factors in single-cells:
The key breakthrough was the idea of fusing reporters to self-assembling peptides, which enables a strong enough signal to be picked up by microscopy while enabling antibody-staining against the peptides to identify which signal matches to a specific reporter. However, SiRIs are not useful for activities that occur in close proximity (i.e. cannot be below micrometers).
Once the group figured out which set of de novo designed peptides generated a strong enough reporter signal in hippocampal neuron cell lines,
In order to analyze how a signal transduction network converts cellular inputs into cellular outputs, ideally one would measure the dynamics of many signals within the network simultaneously used the tool to for a proof-of-concept study of the relationships between calcium signalling, protein kinase A (PKA), and cyclic AMP (cAMP).
The next steps are to use SiRIs in vivo and expand to measuring more than 3 signals. There is a potential to measure up to 16 different things at once in the cell and maybe an animal model.
Genetics, genomics, and developmental biology
Heredity and variation.
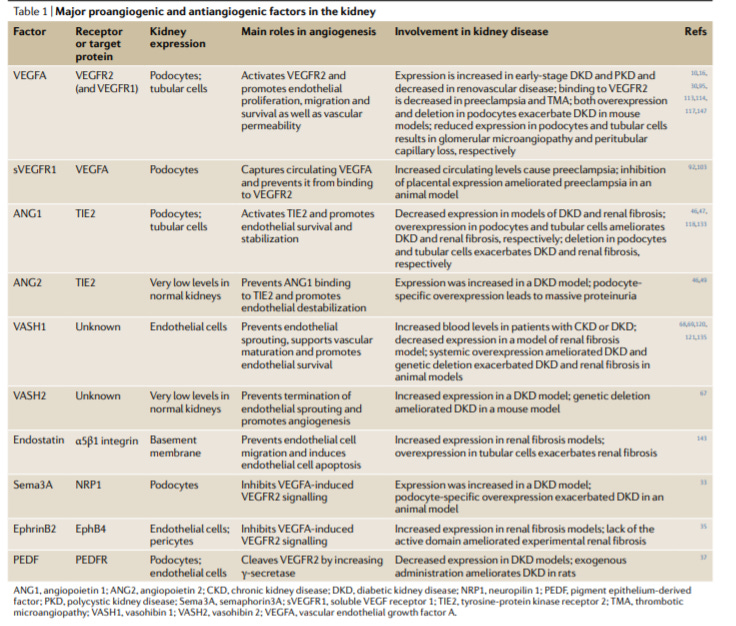
Targeting angiogenesis and lymphangiogenesis in kidney disease - https://www.nature.com/articles/s41581-020-0260-2 - a review from labs at Okayama University Graduate School of Medicine and Tohoku University in Japan did a good job to characterize the role of angiogenesis in kidney disease:
Vascular endothelial growth factor A (VEGF-A) is essential to establish the complex vasculature of the kidney. After development, VEGF-A along with other angiogenic factors maintain the normal structure/function of capillary networks within the kidney.
When the interplay between pro-angiogenic and anti-angiogenic factors is disrupted various kidney diseases emerge
In animal models and some human genetic studies, disruption of VEGF-A activity can lead to glomerular microangiopathy and preeclampsia
While upregulation of VEGF-A drives the development of diabetic kidney disease (DKD) and polycystic kidney disease (PKD)
Angiopoietin 1 and vasohibin 1 have roles in DKD and renal fibrosis
VEGF-C drives lymphangiogenesis and is important in DKD, PKD, and renal fibrosis
However, angiogenesis has different roles in other tissues/organs and targeting these pathways could lead to tumor formation and metastasis. As a result, more selective delivery of medicines to the kidney is required to realize the potential of drugging angiogenesis for renal diseases.
Antisense oligonucleotides (ASO) are a promising modality that has the potential to be kidney selective and are programmable to target several of these angiogenic genes