Axial: https://linktr.ee/axialxyz
Axial partners with great founders and inventors. We invest in early-stage life sciences companies such as Appia Bio, Seranova Bio, Delix Therapeutics, Simcha Therapeutics, among others often when they are no more than an idea. We are fanatical about helping the rare inventor who is compelled to build their own enduring business. If you or someone you know has a great idea or company in life sciences, Axial would be excited to get to know you and possibly invest in your vision and company . We are excited to be in business with you - email us at info@axialvc.com
HSC therapies
Hematopoietic stem cells (HSC) are the source of blood cells from lymphocytes, red blood cells, monocytes among others. HSCs come in two lineages:
Lymphoid - encoding lymphocytes and NK cells
Myeloid - encoding red blood cells, macrophages, and more (this is a large opportunity to make more precise medicines for myeloid-derived diseases since this lineage encodes very essential cells that can lead to toxicity for myeloid-targeting medicines)
Over the last 6 decades, HSC transplants and medicines have become essential to cure several diseases from cancer to blood disorders. The first successful bone marrow derived HSC transplant was performed in the 1950s by E. Donnall Thomas at Fred Hutchinson Cancer Research Center; he won a Nobel Prize for this. Thomas infused bone marrow cells to repopulate the bone marrow of the patient to produce new blood cells. In 1961, the HSC was defined and characterized by two features: (1) self-renewal and (2) can produce different types of blood cells. Simply, resetting the immune system with an HSC transplant is incredibly powerful and curative. The procedure can be divided into two categories:
Allogeneic - hematopoietic stem and progenitor cells (HSPC) are sourced from a healthy donor and used to re-populate another patient’s hematopoietic and immune systems (~40% of transplants)
Autologous - the patient's own HSPC are used for a transplant (~60%)
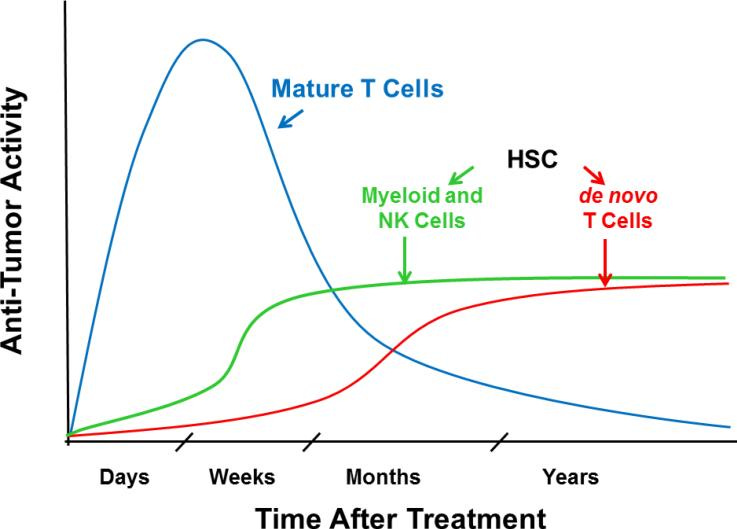
To ensure the transplant engrafts and long-term durability for the patient, the material sourced must have HSCs. Finding healthy and immune compatible HSCs is another issue. As well as the conditioning regimens that are required to reset the immune system and improve the odds of a successful HSC engraftment. As a result, HSC transplants have pretty high mortality rates, which limits its uses in non-malignant/non-life-threatening diseases. However, if these issues can be solved, HSCs have the potential to create long-term cures for immune-related disease. For example, checkpoint inhibitors and CAR-T cell therapies have issues with response rates and sometimes safety. Combining these medicines with HSCs have the potential to lead to strong response rates with durable effects:
Source: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3901057/
However, HSCs face several barriers from becoming more widely used:
HSCs are unable to proliferate and differentiate in vitro. There is a high need for new sources of healthy and immune-compatible HSCs. Umbilical cord blood is a source of a large number of HSCs but has high costs and long timelines for collection, enrichment, HLA typing, and more.
Then determining how many HSCs are in a transplant is pretty difficult. Around 1 out of 10,000 cells in the bone marrow are thought to be HSCs with that number becoming 1 in 100,000 for blood. Developing better biomarkers (i.e. CD34) and sorting tools are needed.
HLA matching is important before a HSC transplant; however, there are low HLA matching rates in the human population. Finding ways to enable successful transplants without HLA matching is important or creating large registries to make matching easier. Figuring this out with minimizing/eliminating graft-versus-host-disease (GvHD) is important. GvHD is probably the biggest barrier for HSCs.
Multiple infusions are often required that can lead to infusion reactions, late toxicities, and relapse in patients.
Expanding and collecting HSCs; developing better harvesting methods and mobilization drugs is valuable.
Engineering HSCs with gene transfers (i.e. viral) and editing may expand the capabilities of an HSC. Ex vivo methods are limited by the small proportion of HSCs in a bone marrow population.
Conditioning regimens like chemotherapy and radiation are used to clear out a patient's blood system to improve the probability of an HSC transplant (i.e. prevent GvHD). Antibodies against targets like CD47 and CD45 have been shown to improve engraftment with less toxicity.
Ultimately, if these problems are solved, HSC medicines have the potential to cure, or at least improve patient mortality rates substantially, most diseases driven by the human immune system.