Axial: https://linktr.ee/axialxyz
Axial partners with great founders and inventors. We invest in early-stage life sciences companies such as Appia Bio, Seranova Bio, Delix Therapeutics, Simcha Therapeutics, among others often when they are no more than an idea. We are fanatical about helping the rare inventor who is compelled to build their own enduring business. If you or someone you know has a great idea or company in life sciences, Axial would be excited to get to know you and possibly invest in your vision and company . We are excited to be in business with you - email us at info@axialvc.com
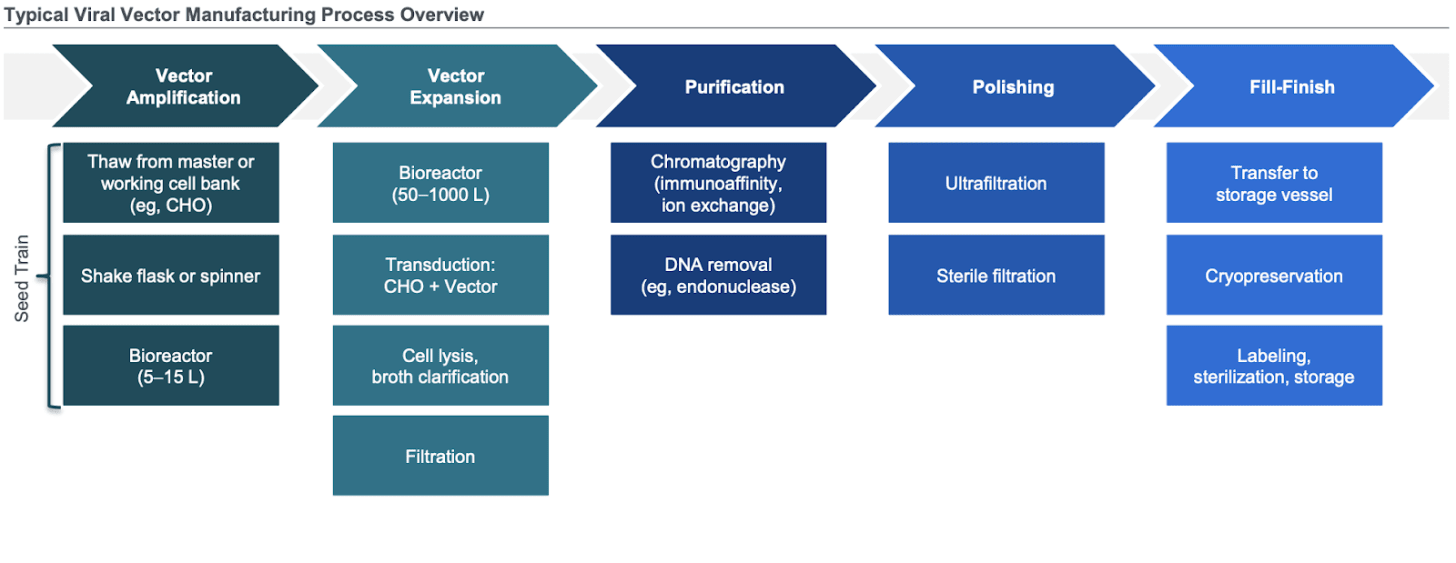
AAV biomanufacturing
Biomanufacturing of adeno-associated virus (AAV) is composed of 5 main steps:
Cell culture (insect cells, HEK293) - HEK293 is the standard
Culture (suspension, 3D adherent, 2D) - suspension
Vector production (transient expression, stable cell line) - transient and takes 2-3 days
Recovery (filtration, centrifugation, TFF) - filtration and TFF
Purification (ultracentrifugation, chromatography) - chromatography
The first AAV gene therapy approved was Glybera in 2012 for LPLD in the EU followed by FDA approval of Luxturna in 2017 for IRD and Zolgensma in 2019 for SMA. As more of these medicines come through the clinic, more manufacturing capacity is required to expand to indications with larger patient populations.
Currently, two methods are the standard for AAV manufacturing - HEK293 and Sf9:
HEK293 cells that are adapted for suspension and are transfected with plasmid DNA. A master cell line is required along with plasmid DNA and transfection agents that increase COGS for each batch. Stoichiometry and expression become important when there are multiple plasmids used (helper and transfer).
Spodoptera frugiperda (Sf9) insect cells using the baculovirus expression vector system (BEVS) for transfection. During a manufacturing run, baculoviruses (one for rep/cap and the other for the AAV genome) with the transfer genes to generate the AAV vectors are infused with the Sf9 cell culture. Each baculovirus requires a master line, which increases the upfront investment required versus HEK292. However, yields in an Sf9 system are significantly higher.
A major theme in gene therapies is that these processes are the actual product. Ultimately, the goal is to develop cell-lines that maximize viral titers (i.e. produce fewer empty capsids) and minimize upfront development costs (a batch can cost upwards of $500K). However, the AAV manufacturing field has 3 major problems to surmount:
Developing stable cell lines to increase yields - the current standard for vector production relies on transient transfection, mainly to reduce costs
Minimize the variability between batches, serotypes, and cargos - depending on the serotype and what is being delivered can lead to wide variations in outcomes across manufacturing runs
Reduce the number of empty capsids produced - cell lines produce many non-functional empty capsids that need to be separated from functional capsids
Improve purification - recovering AAV vectors from culture supernatant versus cell lysate. Moreover, generic processes particularly around purification are needed to apply across multiple vectors.
Over time, there is a large opportunity to design and scale vector-specific manufacturing processes. With more than 12 AAV serotypes, 100s of natural variants, and many more synthetic versions, the same focus and specificity required for drug development is needed in biomanufacturing.