Stress response silencing by an E3 ligase mutated in neurodegeneration
Inventors & their inventions
Axial: https://linktr.ee/axialxyz
Axial partners with great founders and inventors. We invest in early-stage life sciences companies such as Appia Bio, Seranova Bio, Delix Therapeutics, Simcha Therapeutics, among others often when they are no more than an idea. We are fanatical about helping the rare inventor who is compelled to build their own enduring business. If you or someone you know has a great idea or company in life sciences, Axial would be excited to get to know you and possibly invest in your vision and company. We are excited to be in business with you — email us at info@axialvc.com
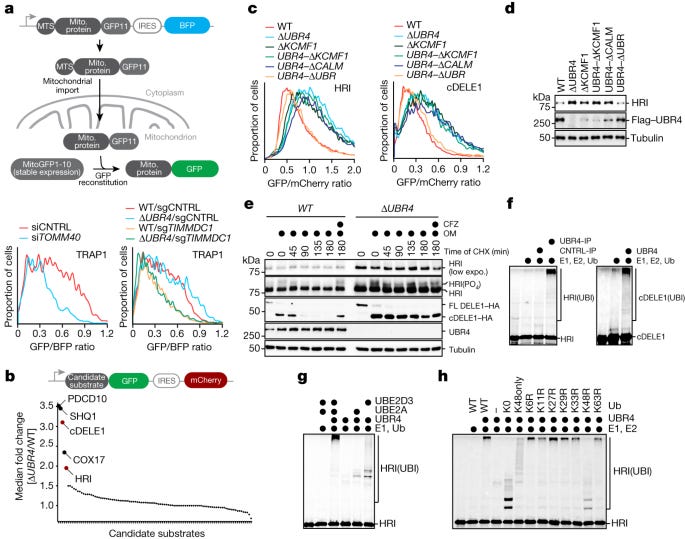
This groundbreaking study investigates the mechanisms by which cells regulate and terminate stress response pathways, particularly in the context of mitochondrial protein import stress. The researchers identify a novel E3 ubiquitin ligase complex they name SIFI (Silencing Factor of the Integrated Stress Response) that plays a crucial role in turning off the cellular stress response after mitochondrial stress has been resolved.
Cells in our bodies are constantly exposed to various stressors, including toxins, temperature fluctuations, and nutrient limitations. To cope with these challenges, cells have evolved conserved signaling pathways known as stress responses. These pathways modulate basic cellular processes to provide time and resources for damage repair. While transient activation of stress responses is beneficial, prolonged activation can lead to cell death and tissue damage. Therefore, it is crucial for cells to silence these stress responses once the stressful conditions have improved. However, the mechanisms underlying this silencing process have remained poorly understood.
The researchers discovered that the E3 ubiquitin ligase UBR4, along with its binding partners KCMF1 and calmodulin, forms a complex (SIFI) that is critical for cell survival during mitochondrial stress. They used a CRISPR-Cas9 synthetic lethality screen to identify genetic interactions with UBR4, finding that cells lacking UBR4 were particularly sensitive to impairment of mitochondrial protein import or electron transport chain function.
Through a series of biochemical and cellular assays, the study demonstrated that SIFI specifically targets two key proteins involved in the mitochondrial stress response for degradation:
cDELE1 - a cleaved form of DELE1 that acts as a sensor of mitochondrial import stress
HRI - A kinase that initiates the integrated stress response (ISR) by phosphorylating eIF2α
The researchers showed that SIFI ubiquitinates these proteins, targeting them for proteasomal degradation. This degradation is specific to the active forms of these proteins, suggesting that SIFI acts to turn off the stress response once it has served its purpose. The study also revealed that SIFI does not prevent initial stress response activation but instead acts to turn off the response once stress has been resolved. This was evidenced by the prolonged activation of ATF4 (a key ISR effector) in cells lacking UBR4 when exposed to mitochondrial stressors. RNA sequencing analysis confirmed that SIFI limits ISR signaling after mitochondrial stress. A particularly novel finding is that SIFI recognizes specific helical motifs in its target proteins that resemble mitochondrial pre-sequences. These motifs, which the authors term "converging degrons," serve a dual functions: they can direct proteins to mitochondria for import and can target proteins for degradation by SIFI when present in the cytoplasm. This discovery provides insight into how SIFI specifically recognizes its targets and links protein localization with stability.
The study revealed that unimported mitochondrial precursor proteins compete with cDELE1 and HRI for recognition by SIFI. This competition mechanism ensures that the stress response remains active until mitochondrial import is restored and precursor proteins are cleared from the cytoplasm. The researchers demonstrated this by showing that increasing the levels of mitochondrial precursors in the cytoplasm protected DELE1 and HRI from degradation. Importantly, the researchers found that mutations in UBR4 are associated with ataxia and early-onset dementia. They demonstrated that the detrimental effects of UBR4 loss could be mitigated by either depleting HRI/DELE1 or by using ISRIB, a small molecule that inhibits the downstream effects of the integrated stress response.
The identification of SIFI highlights that turning off stress responses is not a passive process but rather an actively regulated one. This insight is crucial for understanding how cells maintain homeostasis and prevent the detrimental effects of prolonged stress signaling.
The mechanism by which SIFI recognizes its targets through converging degrons provides an elegant explanation for how cells can specifically terminate the response to mitochondrial stress while maintaining the ability to respond to other types of cellular stress. The discovery of converging degrons that encode both protein localization and stability information is a novel concept in cellular biology. This finding reveals a previously unappreciated level of complexity in how cells manage protein fate and function.
The competition between mitochondrial precursors and stress response proteins for SIFI recognition provides a mechanism for temporal control of the stress response. This ensures that the response remains active only as long as necessary, preventing both premature termination and excessive prolongation of the stress signal. The association of UBR4 mutations with neurodegenerative diseases provides a direct link between defects in stress response regulation and pathological outcomes. This connection offers new perspectives on the etiology of certain neurodegenerative conditions and suggests potential therapeutic approaches. The finding that inhibiting the integrated stress response (using ISRIB or by depleting HRI/DELE1) can rescue cells with defective SIFI function opens up new avenues for therapeutic intervention in diseases caused by mitochondrial import defects or prolonged stress signaling.
The study has significant implications for our understanding and potential treatment of certain neurodegenerative diseases. The researchers found that mutations in UBR4 are associated with ataxia and early-onset dementia. This suggests that defects in stress response silencing may contribute to these conditions. Many neurodegenerative diseases are associated with mitochondrial dysfunction. The findings in this study provide new insights into how cells respond to and recover from mitochondrial stress, which could be relevant to these conditions. The study demonstrates that inhibiting the integrated stress response (using ISRIB or by depleting HRI/DELE1) can rescue cells with defective SIFI function. This suggests a potential therapeutic strategy for treating diseases caused by mutations in UBR4 or other components of the mitochondrial stress response pathway.
While many neurodegenerative diseases are characterized by protein aggregation, this study suggests that persistent stress signaling, rather than protein aggregation itself, may be a key driver of cellular dysfunction in some cases.
The concept of converging degrons and the competition mechanism for stress response silencing are particularly innovative aspects of this work. These findings not only contribute to our fundamental understanding of cellular biology but also open up new avenues for therapeutic intervention in diseases characterized by mitochondrial dysfunction or prolonged stress signaling.
The potential therapeutic implications of this work are especially exciting. The demonstration that pharmacological inhibition of the integrated stress response can rescue cells with defective SIFI function suggests a possible strategy for treating certain neurodegenerative diseases. This approach is particularly promising because it targets the downstream effects of stress signaling rather than trying to eliminate protein aggregates, which has proven challenging in many neurodegenerative conditions.
Further investigation will be needed to fully understand the structural basis of SIFI's target recognition, the potential role of similar mechanisms in other stress response pathways, and the long-term effects of modulating these pathways in vivo. Overall, this paper represents a significant contribution to the fields of cell biology, stress response regulation, and neurodegeneration research. It provides a strong foundation for future studies that could lead to new therapeutic strategies for a range of diseases characterized by mitochondrial dysfunction or aberrant stress signaling.