Axial: https://linktr.ee/axialxyz
Axial partners with great founders and inventors. We invest in early-stage life sciences companies such as Appia Bio, Seranova Bio, Delix Therapeutics, Simcha Therapeutics, among others often when they are no more than an idea. We are fanatical about helping the rare inventor who is compelled to build their own enduring business. If you or someone you know has a great idea or company in life sciences, Axial would be excited to get to know you and possibly invest in your vision and company. We are excited to be in business with you — email us at info@axialvc.com
This perspective paper discusses the potential of protein engineering to overcome the limitations of cytokine therapies for cancer treatment. Cytokines are signaling proteins that play a crucial role in regulating the immune system, making them promising candidates for cancer immunotherapy. However, despite the early FDA approvals of interleukin-2 (IL-2) and interferon-α (IFNα) for cancer treatment over 30 years ago, cytokine therapies have largely failed to achieve significant success in the clinic due to narrow therapeutic windows and dose-limiting toxicities.
The authors attribute these shortcomings to the discrepancy between the localized and regulated manner in which cytokines function endogenously and the systemic, untargeted administration used in most exogenous cytokine therapies to date. Additionally, the ability of cytokines to stimulate multiple cell types, often with paradoxical effects, presents significant challenges for their translation into effective therapies.
To address these issues, the authors propose leveraging protein engineering strategies to control the time, place, specificity, and duration of cytokine signaling, thereby allowing exogenous cytokine therapies to more closely mimic their endogenous exposure profile and unlock their full therapeutic potential. The authors emphasize the importance of spatiotemporal regulation in cytokine engineering, which involves controlling the timing, location, specificity, and duration of cytokine signaling. This concept is central to their perspective and is discussed in the context of various engineering strategies.
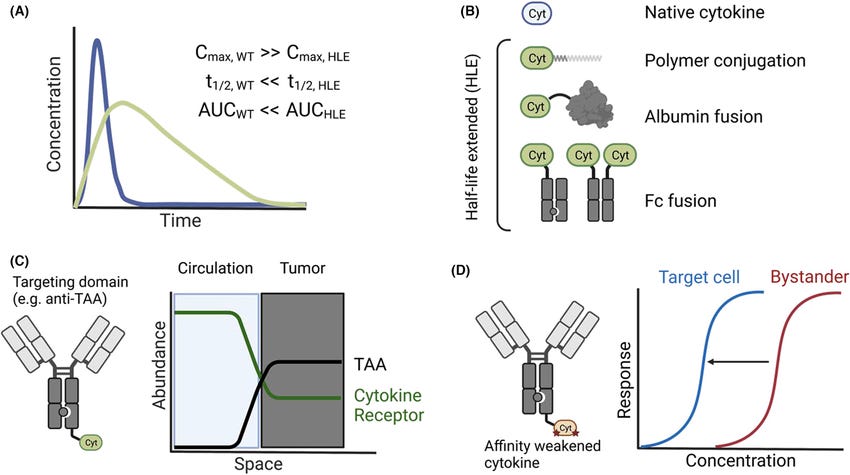
One strategy for temporal programming involves altering the pharmacokinetic properties of cytokines to decrease their maximum serum exposure (Cmax) and extend their half-life (t1/2) and area under the curve (AUC). This can be achieved through half-life extension (HLE) techniques, such as:
1. Fusion to polymers
2. Albumin fusions
3. Antibody Fc domains (often made monovalent through knob-in-hole mutations)
By extending the half-life of cytokines, these strategies aim to reduce the initial peak exposure and maintain a more sustained and tolerable level of cytokine signaling over time.
The authors discuss the limitations of first-generation immunocytokines, which attempted to target cytokines to tumors through tumor-associated antigens (TAAs). However, these efforts were largely unsuccessful due to the minimal solid tumor antigen present in circulation, leading to biodistribution being dominated by the cytokine itself. To address this issue, newer immunocytokine designs utilize affinity-weakened cytokines, where productive signaling is isolated to target cells dictated by an antibody or targeting moiety. This approach achieves effective cell-level spatial programming, enabling more precise targeting of cytokine activity.
The authors use the example of IL-2 engineering to illustrate the challenges and potential strategies for taming cytokine pleiotropy (the ability to affect multiple cell types) through protein engineering. IL-2 signals through the dimeric IL-2Rβγ or trimeric IL-2Rαβγ receptor complexes, with the IL-2Rα (CD25) subunit being constitutively expressed on regulatory T cells (Tregs). A prominent hypothesis in IL-2 engineering has been to develop mutants with biased affinity for the dimeric complex to spare immunosuppressive Tregs, a strategy referred to as "not alpha" IL-2. However, the authors argue that even though "not alpha" candidates ablate CD25 binding, they will still bind Tregs with equivalent affinity to other IL-2Rβγ-expressing cells, such as natural killer (NK) cells, which have the highest abundance of IL-2Rβγ. This suggests that further engineering strategies, beyond receptor biasing, may be necessary to effectively translate IL-2 therapies into the clinic.
The authors' perspective highlights the potential of protein engineering to overcome the limitations of cytokine therapies for cancer treatment. By controlling the spatiotemporal aspects of cytokine signaling, exogenous cytokine therapies can more closely mimic their endogenous counterparts, potentially improving their therapeutic efficacy and safety profiles. The strategies discussed, such as half-life extension, targeted delivery, and receptor biasing, offer promising avenues for further exploration and development. However, as illustrated by the case of IL-2 engineering, a combination of these approaches may be necessary to fully address the complexities of cytokine signaling and achieve effective therapeutic outcomes. Future research efforts should focus on integrating and refining these engineering strategies, as well as exploring new techniques for precise spatiotemporal control of cytokine signaling. Additionally, a deeper understanding of the complex interplay between cytokines and the immune system, as well as the mechanisms underlying their paradoxical effects, will be crucial for informing rational engineering approaches.